Patricia is fifty-seven years old. She came to consult me for fatigue that was not improving despite a normal thyroid panel and correct ferritin levels. Observing her, I noticed a symmetrical skin rash on her décolletage and the back of her hands, dry and rough skin in sun-exposed areas that contrasted with normal skin under clothing. When I asked if these patches had been bothering her for a long time, she told me it was “solar eczema” according to her dermatologist. When I asked if she also had digestive disorders and concentration difficulties, she looked at me with wide eyes: “How did you know?”
Vitamin B3 is perhaps the most fascinating of the B vitamins because it is the precursor to NAD+ (nicotinamide adenine dinucleotide), the most important molecule in cellular energy metabolism. NAD+ is involved in more than five hundred enzymatic reactions. Without it, no Krebs cycle, no respiratory chain, no ATP. No energy. No life.
Causes of B3 deficiency
Niacin has a unique characteristic among vitamins: it can be synthesized by the body from tryptophan, an essential amino acid. The conversion pathway (kynurenine pathway) transforms sixty milligrams of tryptophan into one milligram of niacin, which means that B3 requirements depend on both direct niacin intake and tryptophan intake. This duality explains why populations whose diet is based on corn (poor in tryptophan and whose niacin is bound and not bioavailable) develop pellagra.
Chronic alcoholism is the most common cause of B3 deficiency in industrialized countries. Ethanol diverts NAD+ from its normal metabolic functions for its own catabolism (alcohol dehydrogenase and aldehyde dehydrogenase consume massive amounts of NAD+), and it reduces intestinal absorption of niacin.
Diets low in animal protein limit both direct niacin intake and tryptophan intake. The tryptophan-niacin conversion requires iron, B2, B6, and copper as cofactors, which means that deficiencies in these micronutrients block endogenous B3 production.
Certain medications are major antagonists. Isoniazid (antituberculous) blocks the conversion of tryptophan to niacin and is a classic cause of iatrogenic pellagra. 5-Fluorouracil, mercaptopurine, and other chemotherapies increase NAD+ requirements. Anticonvulsants (phenytoin, carbamazepine) interfere with niacin metabolism.
Carcinoid syndrome diverts tryptophan toward excessive serotonin synthesis by the tumor, depriving the kynurenine pathway of its substrate and creating B3 deficiency. Hartnup disease, a rare genetic disorder of tryptophan transport, can also cause deficiency.
Symptoms of deficiency
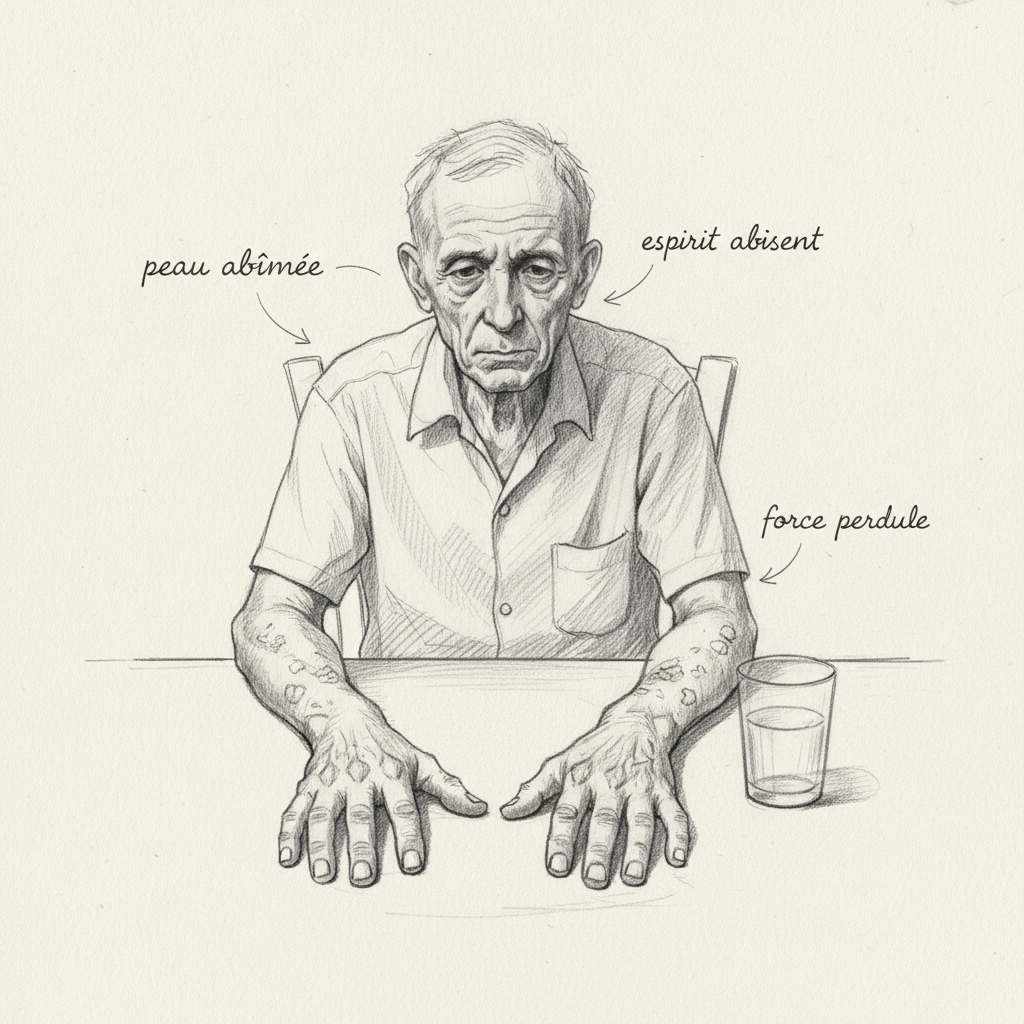
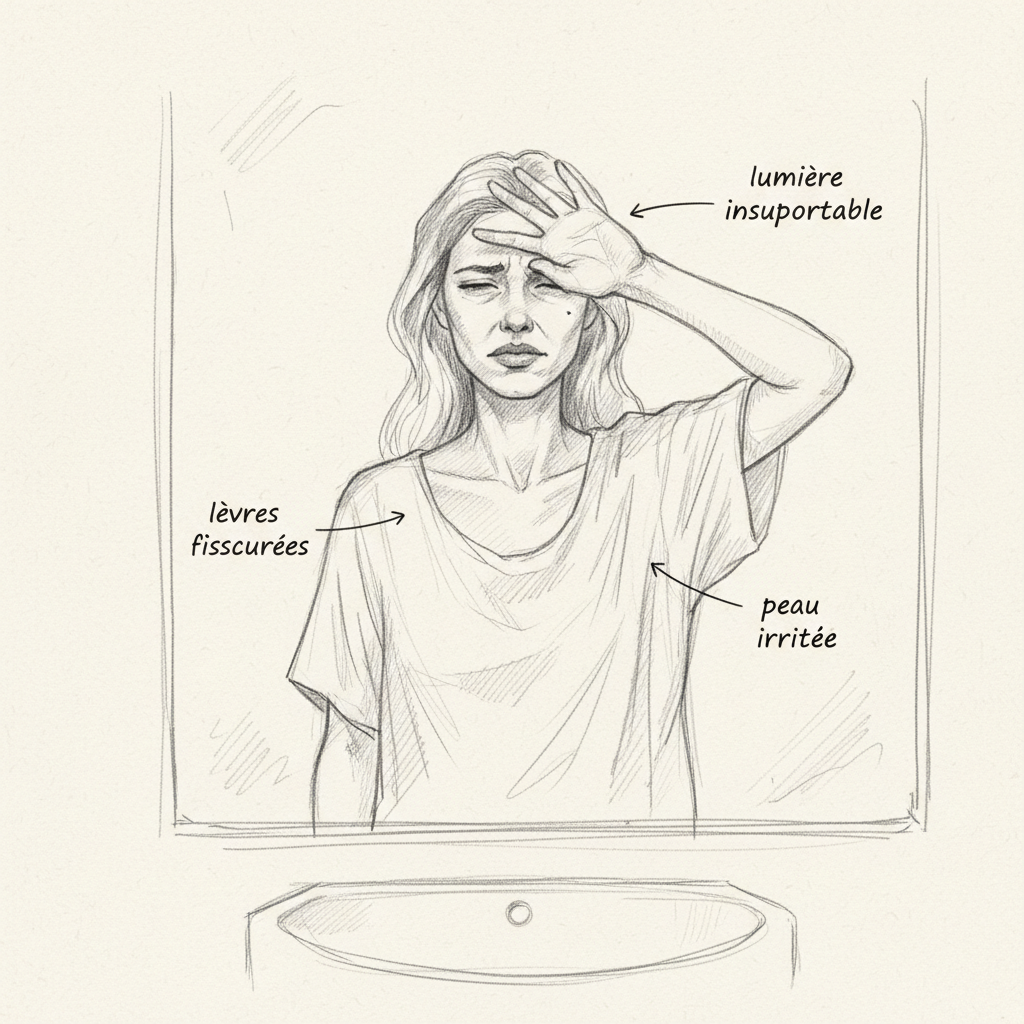
Clinical pellagra is the disease of the three Ds: dermatitis, diarrhea, dementia. Pellagrous dermatitis is pathognomonic: a symmetrical, erythematous then hyperpigmented rash, limited to sun-exposed areas (hands, forearms, V-shaped décolletage or Casal collar, face). Photosensitivity is the cardinal sign: sun causes intense inflammatory reaction on skin deficient in NAD+ that can no longer repair UV damage.
Diarrhea reflects damage to the digestive mucosa whose rapid renewal requires a lot of NAD+. Glossitis (red and painful tongue), nausea, and abdominal pain often precede diarrhea.
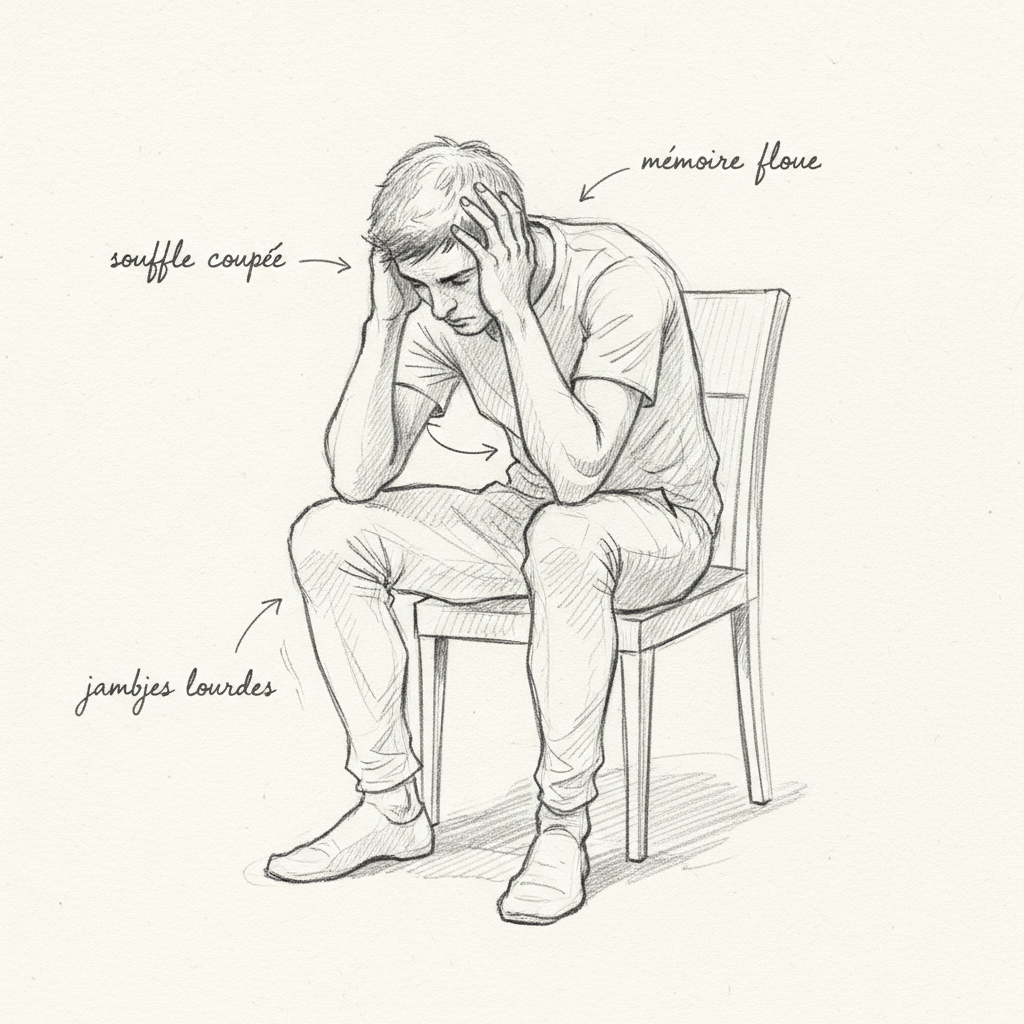
Pellagrous dementia begins with nonspecific psychiatric symptoms: irritability, anxiety, insomnia, concentration difficulties, apathy. It can progress to mental confusion, hallucinations, disorientation, and frank psychosis. The subclinical form, with cognitive fatigue and mood disorders without clear cutaneous and digestive signs, is probably much more common than classic pellagra.
Chronic fatigue is a nearly constant symptom of B3 deficiency because NAD+ is the central coenzyme of mitochondrial ATP production. When NAD+ is lacking, every cell in the body runs in slow motion. It is a deep, cellular fatigue that rest does not relieve.
Micronutrients essential to B3
Tryptophan is the first partner since it is the endogenous precursor of niacin. Adequate tryptophan intake (found in turkey, eggs, cashews, pumpkin seeds, cheese, fish) reduces the need for preformed niacin.
Vitamin B2 (riboflavin) is a cofactor of kynurenine monooxygenase, an enzyme in the tryptophan-niacin pathway. Vitamin B6 is a cofactor of kynurenase in the same pathway. Iron is a cofactor of tryptophan 2,3-dioxygenase, the first enzyme in the pathway. Copper also plays a role in this cascade. Without these four cofactors, even generous tryptophan intake does not produce sufficient niacin.
Zinc is involved in NAD+ metabolism at the level of poly-ADP-ribose polymerases (PARP), DNA repair enzymes that consume NAD+. High oxidative stress activates PARPs, which massively consume NAD+ and create a functional deficit even with adequate B3 intake.
Food sources
Poultry liver is the richest source with 16 milligrams per 100 grams. Fresh tuna provides 15 milligrams per 100 grams. Chicken and turkey contain 10 to 13 milligrams per 100 grams. Salmon provides 8 milligrams per 100 grams. Peanuts contain 14 milligrams per 100 grams, making them the best plant source. Shiitake mushrooms provide 4 milligrams per 100 grams. Sunflower seeds contain 7 milligrams per 100 grams. Legumes (lentils, chickpeas) provide 1.5 to 2 milligrams per 100 grams. Brewer’s yeast contains 40 milligrams per 100 grams.
Recommended intakes are 14 milligrams of niacin equivalents (NE) per day for women and 16 milligrams for men. One niacin equivalent corresponds to 1 milligram of niacin or 60 milligrams of tryptophan. The therapeutic dose for lipid profile is 1500 to 3000 milligrams per day of niacin under medical supervision (liver transaminases must be monitored).
Antagonists of vitamin B3
Alcohol is the main antagonist by diverting NAD+ toward ethanol catabolism. Excess leucine (unbalanced high-protein diets) inhibits tryptophan-to-niacin conversion. Non-nixtamalized corn contains bound niacin (niacytin) that is not bioavailable, which explains endemic pellagra in Africa and Latin America where corn is the staple food without the traditional alkaline treatment of the Aztecs.
Isoniazid is the most powerful medication antagonist. Azathioprine, 6-mercaptopurine, and L-dopa also interfere with B3 metabolism. Chronic oxidative stress depletes NAD+ through PARP hyperactivation.
Forgotten causes of deficiency
Aging is accompanied by progressive decline in intracellular NAD+ levels. Brenner’s studies (2017) showed that NAD+ levels decrease by fifty percent between thirty and sixty years in certain tissues. This NAD+ decline is involved in mitochondrial decline, decreased sirtuin activity (NAD+-dependent longevity proteins), and acceleration of cellular aging. Nicotinamide riboside (NR) is being studied as an anti-aging strategy precisely because it restores NAD+ levels.
Chronic inflammation activates PARPs that massively consume NAD+ to repair DNA damaged by free radicals. A patient with autoimmune disease, chronic intestinal inflammation, or high oxidative stress can have considerably increased B3 requirements.
Intense sun exposure without protection increases NAD+ requirements for repair of skin DNA damaged by UV rays. This is why pellagra is a disease of sunny regions in malnourished populations.
Crohn’s disease and ileal resection reduce absorption of niacin and tryptophan. Liver insufficiency impairs the conversion of tryptophan to NAD+.
Dietary supplements
Niacin (nicotinic acid) is the classic form, effective for lipid profile but responsible for cutaneous flush. Start at 100 milligrams per day and increase gradually to target dose (500 to 2000 milligrams). The flush decreases with regular use. Extended-release niacin reduces flushing but increases hepatotoxicity risk and is not recommended.
Nicotinamide (niacinamide) is the flush-free form, well tolerated, effective for restoring NAD+ but without effect on lipids. The usual dose is 500 to 1500 milligrams per day.
Nicotinamide riboside (NR) is the newest and most promising form for boosting intracellular NAD+. The Trammell study published in Nature Communications in 2016 showed that 1000 milligrams of NR per day increased blood NAD+ two and a half times in healthy adults. NR is the preferred form for aging, mitochondrial fatigue, and neuroprotection.
Nicotinamide mononucleotide (NMN) is another direct NAD+ precursor, intermediate between NR and NAD+ in the synthesis pathway. Animal studies by Yoshino (2011) showed promising results on glucose metabolism and aging.
Patricia started with 500 milligrams of nicotinamide per day, combined with a B complex and magnesium. Within six weeks, her fatigue had significantly improved, her photosensitive dermatitis had nearly disappeared, and her cognitive disorders had faded. The “solar eczema” rash was not eczema at all: it was the subclinical Casal collar of mild pellagra that no one had recognized.
To assess your B3 vitamin status, take the B3 deficiency questionnaire on my site.
To go further
- Carnitine and thyroid: the molecule no one tests
- Vitamin B1 (thiamine): the spark of your energy and your brain
- Vitamin B2 (riboflavin): your mitochondria don’t run without it
- Vitamin B5 (pantothenic acid): the vitamin of your adrenals and coenzyme A
Sources
- Brenner, Charles. “Boosting NAD+ with a small molecule that activates NAMPT.” Nature Chemical Biology 13 (2017): 1125-1126.
- Trammell, Samuel A. J., et al. “Nicotinamide riboside is uniquely and orally bioavailable in mice and humans.” Nature Communications 7 (2016): 12948.
- Yoshino, Jun, et al. “Nicotinamide mononucleotide, a key NAD+ intermediate, treats the pathophysiology of diet- and age-induced diabetes in mice.” Cell Metabolism 14.4 (2011): 528-536.
- Curtay, Jean-Paul. Nutrithérapie: bases scientifiques et pratique médicale. Testez Éditions, 2016.
- Mouton, Georges. Écologie digestive. Marco Pietteur, 2004.
If you want personalized support in micronutrition, you can schedule a consultation. I consult in my office in Paris and by video throughout France.
For supplementation, Sunday Natural offers B vitamins and pharmaceutical-quality nicotinamide riboside (ten percent off with code FRANCOIS10). Find all my partnerships with exclusive promo codes.



Laisser un commentaire
Sois le premier à commenter cet article.