Thomas is fifty-two years old. He eats organic, does sport three times a week, doesn’t smoke, drinks moderately. For two years, he’s been taking an antioxidant supplement bought at a pharmacy: vitamin C, vitamin E, beta-carotene, the classic trio sold by the millions with the promise of “fighting aging.” Two years later, Thomas is still tired. His joints hurt when he wakes up. His skin has lost its luster. His standard blood tests are normal. And when I prescribed him a complete oxidative assessment, the result was a shock: his erythrocyte SOD was collapsed, his glutathione peroxidase was low, his selenium was at 65 µg/L (target > 100), and his reduced glutathione was at its lowest point. Thomas had been taking antioxidants for two years, and yet his body was in the throes of oxidative stress.
Thomas’s paradox illustrates a fundamental error that Prof. Vincent Castronovo, MD PhD, professor at the University of Liège and instructor in the DU in Micronutrition, breaks down in his course on energy and oxidative stress. Taking exogenous antioxidants when endogenous defenses are collapsed is like repainting a facade whose foundations are crumbling. Thomas’s problem wasn’t a lack of vitamin C. It was a deficiency in selenium, zinc, and glutathione cofactors that prevented his antioxidant enzymes from functioning.
“Our body defends itself against the destructive activity of free radicals through safe and effective antioxidant systems. If free radical production increases beyond defense capabilities, or if these defenses are defective, then free radicals will be able to attack our own molecules.” Prof. Vincent Castronovo, DU MAPS 2020, course #13 “Energy and Oxidative Stress”1
Your cells burn 50 kilos of ATP per day
To understand oxidative stress, you must first understand where your cells’ energy comes from. An individual burns approximately 50 kilos of ATP per day2. Fifty kilos. ATP (adenosine triphosphate) is the universal energy currency of life. Every muscle contraction, every heartbeat, every nerve transmission, every protein synthesis consumes ATP.
This ATP is produced primarily in mitochondria, cellular organelles that are actually ancient bacteria. 1.6 billion years ago, they invented a biochemical system to use the initially toxic properties of oxygen: cellular respiration. This invention multiplied energy production by a factor of 18 compared to anaerobic fermentation.
We possess approximately 3 quadrillion mitochondria (3,000,000,000,000,000)3. Three thousand million millions. The heart, brain, liver, and muscles are the organs richest in mitochondria because they consume the most energy. The mitochondrion has its own DNA (16,569 base pairs, transmitted exclusively by the mother), its own ribosomes, and its own protein synthesis machinery. It is literally an organism within an organism.
The electron transport chain, housed in the inner mitochondrial membrane, is the central mechanism of ATP production. Electrons extracted from nutrients (glucose via glycolysis, fatty acids via beta-oxidation) are transported by NAD (whose precursor is vitamin B3) and FAD (whose precursor is vitamin B2) through a series of protein complexes (complexes I through V)4. The energy released by this transfer pumps protons (H+) into the intermembrane space, creating a gradient that spins ATP synthase (complex V), the molecular turbine that assembles ATP.
Coenzyme Q10 is the electron carrier between complex I and complex II. Cytochrome C takes over when the electron has lost some of its energy. Iron and copper are essential cofactors of complexes I, II, III, and IV. The article on cellular aging details in depth the link between mitochondria, energy, and aging.
10% of electrons fall: the source of oxidative stress
Here is the crucial point. The electron transport chain is not perfect. Approximately 10% of highly energetic electrons “fall” from the transport chain before reaching oxygen in a controlled way5. These fugitive electrons meet molecular oxygen (O2) uncontrollably and transform it into superoxide radical (O2•-), an extremely unstable and toxic reactive oxygen species (ROS).
This is the price we pay for breathing. Oxygen is both our fuel and our poison. Every breath produces energy AND free radicals. It is a Faustian bargain that evolution accepted 1.6 billion years ago in exchange for the energetic power of aerobic respiration.
Free radicals don’t come only from mitochondria. We distinguish three main endogenous sources: the mitochondrion (major source), inflammation (activation of macrophages and neutrophils that produce ROS to kill pathogens), and hepatic detoxification (phase I of cytochrome P450 generates reactive intermediates)6. Exogenous sources include UV rays, ionizing radiation, tobacco, alcohol, air pollutants, pesticides, and xenobiotics.
The targets of free radicals are three categories of biological molecules. Proteins are denatured by oxidation of their thiol groups, which alters enzymes and receptors. Lipids, particularly polyunsaturated fatty acids in cell membranes, undergo chain peroxidation that stiffens membranes and disrupts mitochondrial function (whose inner membrane is rich in DHA)7. DNA suffers single and double-strand breaks, base modifications (8-OHdG), and mutations that accumulate with age and promote carcinogenesis.
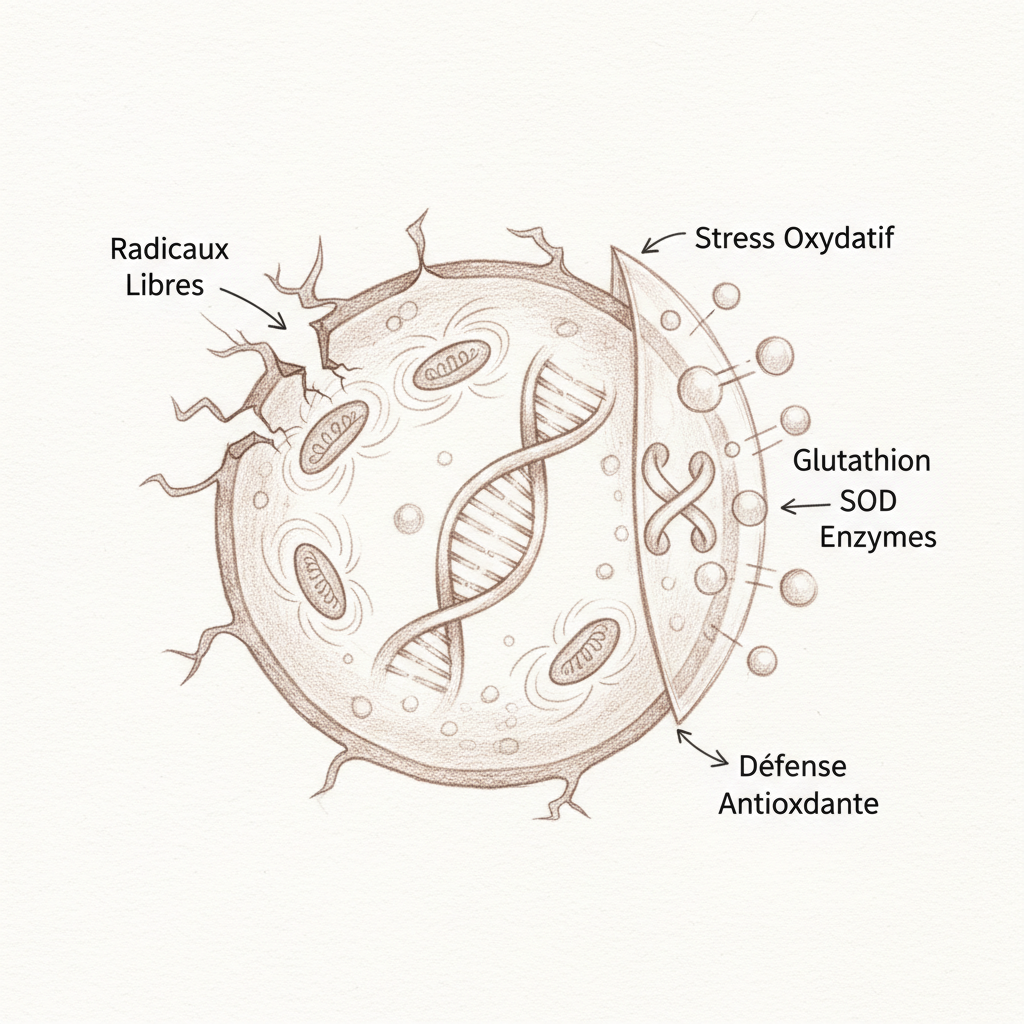
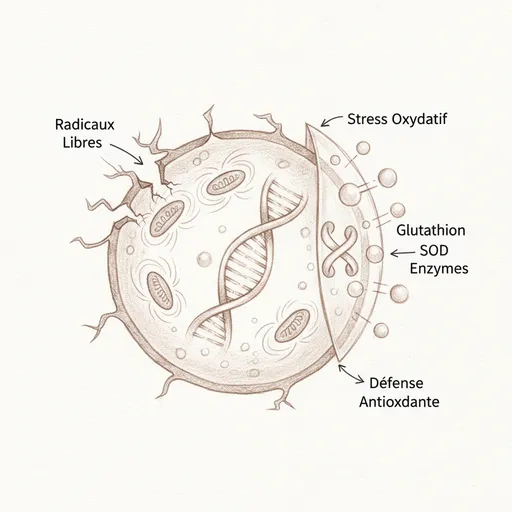
The triad of defense: SOD, GPx, catalase
Our body is not without defense. The endogenous antioxidant defense system is remarkably sophisticated, organized in three lines8.
The first line is superoxide dismutase (SOD). It transforms the superoxide radical (O2•-) into hydrogen peroxide (H2O2). There are three forms of SOD: SOD1 (copper-zinc), located in the cytoplasm, SOD2 (manganese), located in the mitochondrial matrix, and SOD3 (copper-zinc), extracellular. Copper, zinc, and manganese are therefore direct cofactors of the first line of antioxidant defense. A zinc deficiency directly weakens this first barrier.
The second line is glutathione peroxidase (GPx). It transforms hydrogen peroxide (H2O2) into water (H2O). Its essential cofactor is selenium. Without selenium, GPx doesn’t function, and H2O2 accumulates in the cell. Reduced glutathione (GSH) is the electron donor of GPx: it is oxidized to glutathione disulfide (GSSG) during the reaction, then regenerated by glutathione reductase (which requires NADPH, itself derived from the pentose phosphate pathway)9. Catalase, present mainly in liver peroxysomes, constitutes a complementary pathway for H2O2 degradation.
The catastrophe occurs when H2O2 is not neutralized. In the presence of ferrous iron (Fe2+) or copper (Cu+), H2O2 spontaneously transforms into hydroxyl radical (OH•) via the Fenton reaction. And that’s when everything shifts: we have no enzymatic system capable of neutralizing the hydroxyl radical10. The OH• radical is the most destructive of all free radicals. It is a molecular bomb that attacks everything in its path within a few nanometers. When produced, damage is inevitable.
This is why excess free iron is so dangerous. This is why ferritin must be monitored not only for anemia (low ferritin) but also for overload (ferritin > 200 in women, > 300 in men). This is why excess free copper is pro-oxidant and the copper/zinc ratio must be measured. And this is why selenium is probably the most important and most underestimated antioxidant micronutrient.
Selenium: the guardian that 30% of French people don’t have
An alarming figure: 30% of French people have a selenium deficiency11. Thirty percent. That’s one in three French people whose glutathione peroxidase cannot function properly, whose H2O2 accumulates, and whose risk of Fenton reaction increases.
The Brooks et al. (2001) study cited in the course is particularly striking. Published in the Journal of Urology, it shows that low plasma selenium levels before diagnosis are associated with a 4 to 5 times higher risk of prostate cancer12. Men in the lowest quartile of selenium had a risk multiplied by five compared to those in the highest quartile. Selenium decreases significantly with age, making supplementation particularly important in men over 50.
Selenium does not act alone. It is a cofactor not only of GPx but also of thioredoxin reductases and iodothyronine deiodinases (the enzymes that convert T4 to active T3 in the thyroid). A selenium deficiency therefore simultaneously affects antioxidant defense AND thyroid function, which explains the frequency of Hashimoto + oxidative stress associations.
Alpha-lipoic acid: the universal antioxidant
One antioxidant deserves the title of “first antioxidant of the mitochondria” and “universal antioxidant”: alpha-lipoic acid13.
Alpha-lipoic acid possesses unique properties. It is both water-soluble and fat-soluble, which allows it to act in all cellular compartments. It crosses the blood-brain barrier (protecting the central nervous system). It neutralizes many different free radicals. And most importantly, it regenerates all other antioxidants after they have been oxidized: vitamin C, vitamin E, glutathione, thioredoxin, and ubiquinone (CoQ10). In its reduced form (dihydrolipoic acid), it is the chief recycler of the antioxidant network.
It is also the primary cofactor of pyruvate decarboxylase, the enzyme complex that allows pyruvate to enter the mitochondrion (with vitamin B1). Without alpha-lipoic acid, glucose cannot be converted to mitochondrial energy. This is why it is particularly studied in type 2 diabetes and diabetic neuropathy.
Exogenous antioxidants: complements, not substitutes
Exogenous antioxidants are complements to endogenous defenses, not substitutes14.
Vitamin E (tocopherols and tocotrienols) protects cell membranes against lipid peroxidation. Vitamin C protects aqueous compartments and regenerates oxidized vitamin E. Carotenoids (beta-carotene, lycopene, lutein, zeaxanthin) protect membranes and skin against UV rays. Lycopene from tomatoes is the most powerful carotenoid thanks to its long chain of conjugated double bonds. Polyphenols (flavonoids, resveratrol, curcumin, green tea catechins) act as antioxidants but also as modulators of gene expression via the NRF2 and NF-κB pathways.
The fundamental principle: “different antioxidants for different cellular compartments.” Vitamin E protects lipid membranes. Glutathione and selenium protect the cytoplasm. Manganese SOD protects the mitochondrial matrix. Alpha-lipoic acid crosses all compartments. Taking a single antioxidant hoping to protect the entire cell is like posting a single guard at a castle with a hundred doors.
Mitochondrial micronutrients: the complete list
Here is the complete list of micronutrients necessary for optimal mitochondrial function15:
- Vitamins B1 (thiamine), B2 (riboflavin), B3 (niacin), B5 (pantothenic acid): cofactors of ATP production pathways
- Iron and copper: cofactors of respiratory chain complexes
- Zinc and manganese: cofactors of SOD
- Selenium: cofactor of GPx
- Alpha-lipoic acid: mitochondrial antioxidant and cofactor of pyruvate decarboxylase
- Coenzyme Q10: electron carrier between complexes I and III
- Reduced glutathione: master intracellular antioxidant
- L-carnitine: transporter of fatty acids across the mitochondrial membrane
- Omega-3 (DHA): essential component of the inner mitochondrial membrane (fluidity)
- Vitamin E and vitamin C: complementary exogenous antioxidants
- Vitamin A: regulation of mitochondrial biogenesis
- L-glutamine: energy substrate for enterocytes and lymphocytes
The story of migratory birds beautifully illustrates the role of omega-3. The semipalmated sandpiper (Calidris pusilla) doubles its body mass in two weeks by feeding on shrimp rich in EPA and DHA before a direct flight over the Atlantic. Omega-3s incorporate into the membranes of its locomotor muscles, increase membrane fluidity, stimulate mitochondrial biogenesis, and improve aerobic capacity16. It’s not just stored energy, it’s a metabolic signal that prepares muscles for endurance.
The mitochondrial vicious cycle
There is a vicious cycle at the heart of mitochondrial medicine. Mutations in mitochondrial DNA (10 times more fragile than nuclear DNA because it’s not protected by histones and equipped with archaic repair mechanisms) alter proteins in respiratory complexes. Defective complexes drop more electrons. More dropped electrons means more free radicals. More free radicals cause further mitochondrial DNA damage. And the cycle accelerates with age17.
This vicious cycle is implicated in liver disease, diabetes, obesity, cardiovascular disease, cancer, Alzheimer’s, autism, chronic fatigue, and accelerated aging. The mitochondrial theory of aging, developed by Harman as early as 1972 and confirmed by Lenaz (1999), considers somatic mutations in mitochondrial DNA induced by free radicals as the primary cause of age-related energy decline18.
An optimal mitochondrial nutritional environment is a prerequisite for preserving health and ensuring harmonious aging. Mitochondrial health is not a niche topic for biologists. It is the foundation of all health.
Naturopathic protocol against oxidative stress
Here is the three-axis protocol I use in consultation after a complete oxidative assessment.
Axis 1: Restore endogenous defenses. This is the absolute priority. Selenium 100 to 200 µg/day (selenomethionine). Zinc bisglycinate 15 to 25 mg/day (SOD1 cofactor). Manganese 2 to 5 mg/day (SOD2 cofactor). NAC 600 to 1200 mg/day (glutathione precursor). Alpha-lipoic acid 300 to 600 mg/day (regenerator of all antioxidants). CoQ10 ubiquinol 100 to 200 mg/day (especially if on statins, fatigue, cardiomyopathy). Magnesium is an indirect cofactor (MgATP, SOD).
Axis 2: Provide complementary exogenous antioxidants. Vitamin C 500 to 1000 mg/day (divided in 2 doses). Natural vitamin E (mixture of tocopherols, not isolated alpha-tocopherol) 200 to 400 IU/day. Mixed carotenoids (lycopene, lutein, natural beta-carotene) via colorful eating. Polyphenols: curcumin (200 to 500 mg/day), resveratrol, green tea EGCG, quercetin.
Axis 3: Reduce sources of free radicals. Limit exposure to xenobiotics. Manage chronic stress (source of inflammation and ROS). Practice moderate physical activity (sport at moderate intensity increases endogenous defenses; overtraining depletes them). Treat low-grade chronic inflammation. Correct iron deficiencies (anemia) and monitor iron excess (overload). Favor gentle cooking (high-temperature cooking generates pro-oxidant Maillard products).
When to consult and limitations of the approach
Thomas, after four months of targeted correction based on his oxidative assessment, saw his markers normalize. His selenium rose from 65 to 112 µg/L. His GPx normalized. His reduced glutathione increased. And most importantly, his fatigue disappeared, his joint pain melted away, and his vitality returned. He still takes antioxidants, but not those from the supermarket. He takes selenium, NAC, alpha-lipoic acid, and CoQ10. His foundations are restored. The facade followed suit.
The oxidative assessment is a powerful tool but it requires a specialized laboratory and interpretation by a trained practitioner. The markers must be interpreted together, not in isolation. Excess SOD with low GPx indicates that the first line of defense is working but the second is failing (selenium or glutathione deficiency). Excess TBARS with normal defenses indicates an exogenous source of free radicals (tobacco, pollution, medications). Each profile is unique and correction must be individualized.
The limitation of this approach is that oxidative stress is never isolated. It is linked to inflammation, insulin resistance, dysbiosis, toxic burden, and chronic stress. The complete assessment of the 7 pillars of nutritional medicine is often necessary to understand the full picture. But if you had to retain one thing from this article, remember this: your cells need selenium, zinc, glutathione, and CoQ10 to defend themselves. Not vitamin C in an effervescent tablet.
To go further
- Oxidative balance: Dr. Brack’s test to measure your oxidative stress
- Marchesseau’s oxidative balance: free radicals, aging, and antioxidant defenses
- Cancer and nutrition: what micronutrition changes in the equation
- Cholesterol and cardiovascular disease: the real culprits your cardiologist isn’t looking for
Sources
Footnotes
-
Castronovo V. DU MAPS 2020, course #13 “Energy Metabolism: Oxidative Stress”. Slide: “Safe and effective antioxidant systems.” ↩
-
Castronovo V. DU MAPS 2020, course #13. Slide: “An individual burns approximately 50 kilos of ATP per day.” ↩
-
Castronovo V. DU MAPS 2020, course #13. Slide: “3 quadrillion mitochondria per individual.” ↩
-
Castronovo V. DU MAPS 2020, course #13. Slides: “NAD: vitamin B3; FAD: vitamin B2. Electron transport chain.” ↩
-
Castronovo V. DU MAPS 2020, course #13. Slide: “10% of highly energetic electrons fall from the transport chain.” ↩
-
Castronovo V. DU MAPS 2020, course #13. Slides: “Free radicals of endogenous origin: mitochondrion, inflammation, detoxification.” ↩
-
Castronovo V. DU MAPS 2020, course #13. Slide: “Membrane fluidity and DHA: natural doping in migratory birds.” ↩
-
Castronovo V. DU MAPS 2020, course #13. Slides: “SOD (Cu,Zn / Mn) → GPx (Se, GSH) → Catalase. Fenton reaction.” ↩
-
Castronovo V. DU MAPS 2020, course #13. Slide: “Glutathione Peroxidase System. Any selenium deficiency causes H2O2 accumulation.” ↩
-
Castronovo V. DU MAPS 2020, course #13. Slide: “We have NO enzymatic system to destroy the OH• radical. Fenton reaction.” ↩
-
Castronovo V. DU MAPS 2020, course #13. Slide: “30% of French people have a selenium deficiency.” ↩
-
Brooks JD et al. Plasma selenium level before diagnosis and the risk of prostate cancer development. J Urol. 2001;166(6):2034-8. Cited in Castronovo slide. ↩
-
Castronovo V. DU MAPS 2020, course #13. Slides: “Alpha-lipoic acid: The Universal Antioxidant. Regenerates vitamin C, E, glutathione, thioredoxin, ubiquinone.” ↩
-
Castronovo V. DU MAPS 2020, course #13. Slide: “Different antioxidants for different cellular compartments.” ↩
-
Castronovo V. DU MAPS 2020, course #13. Slide: “Mitochondrial micronutrients: complete list.” ↩
-
Weber JM. The physiology of long-distance migration: extending the limits of endurance metabolism. J Exp Biol. 2009;212:593-597. Cited in Castronovo slide. ↩
-
Castronovo V. DU MAPS 2020, course #13. Slide: “Mitochondrial DNA 10 times more fragile. Vicious cycle mutations → ROS → mutations.” ↩
-
Lenaz G et al. Mitochondria, oxidative stress, and antioxidant defences. Acta Biochim Pol. 1999;46(1):1-21. Cited in Castronovo slide. ↩

Laisser un commentaire
Sois le premier à commenter cet article.