Vitamin A: The Forgotten Vitamin
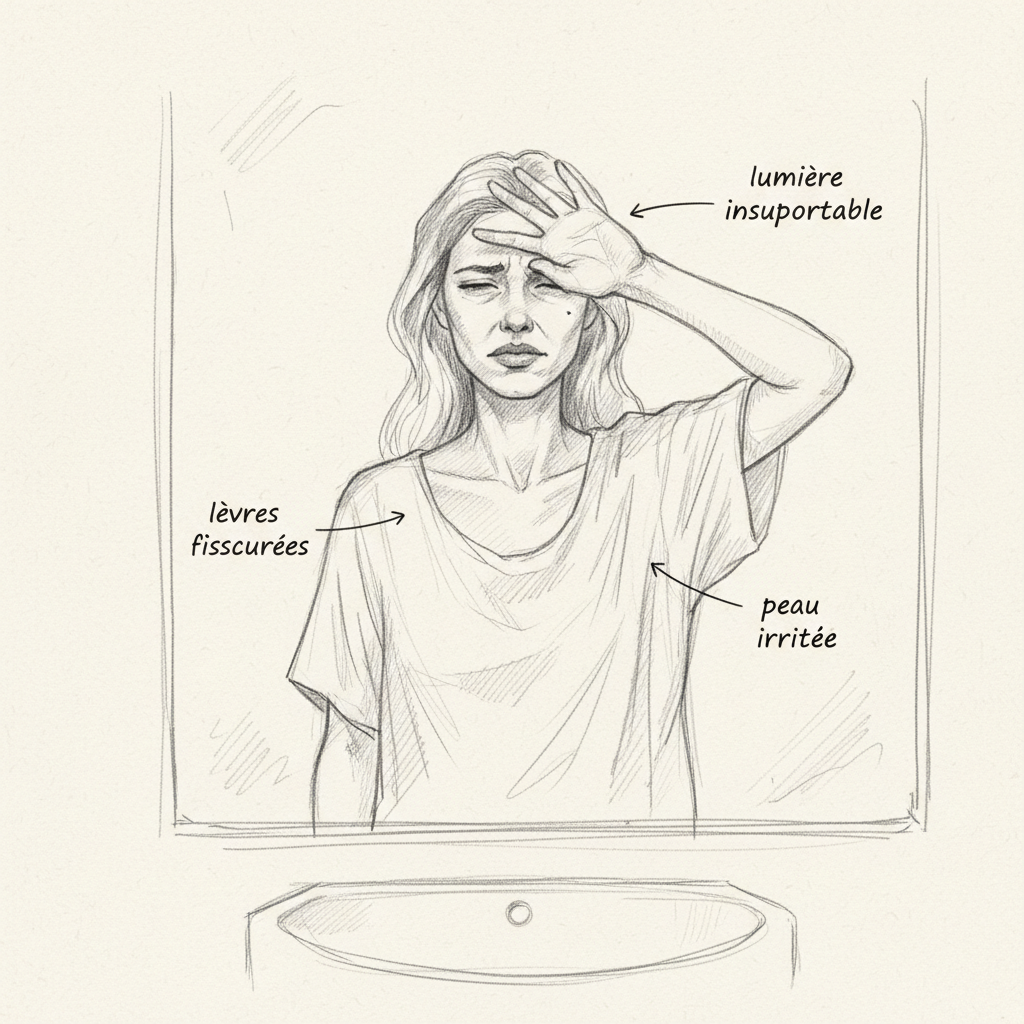
Nathalie is forty-five years old. She came to consult me for chronic fatigue that her doctor attributed to stress, dry skin that her dermatologist treated with moisturizing cream, and recurrent ENT infections for which she was prescribed antibiotics every two months. By digging deeper into her history, one detail put me on the right track: for about a year, she had difficulty driving at night. The headlights of oncoming cars dazzled her more, and especially, she took an abnormally long time to recover her vision after being dazzled. “My ophthalmologist told me it was normal for my age.” No, it’s not normal at forty-five. It’s a classic warning sign of vitamin A deficiency, and old-school doctors knew this.
Looking at her labs more carefully, I found that she had been taking a proton pump inhibitor for three years for acid reflux, had undergone cholecystectomy (removal of the gallbladder) five years ago, and ate very little animal fat out of fear of cholesterol. Three factors converging on the same result: malabsorption of fat-soluble vitamins, including vitamin A. Her liver, deprived of properly concentrated bile, could no longer absorb or store retinol. Her skin, mucous membranes, eyes, and immune system had been paying the price for months.
“The terrain is everything, the microbe is nothing.” Antoine Béchamp
Vitamin A is the great forgotten nutrient in modern micronutrition. Everyone talks about vitamin D, magnesium, zinc. But vitamin A, the one without which you can’t see in the dark, the one that lines and protects every mucous membrane in your body, the one that allows your lymphocytes to differentiate properly, is systematically ignored in nutritional assessments. Yet the World Health Organization considers vitamin A deficiency as the leading cause of preventable blindness worldwide, and a major factor in child mortality from infections in developing countries1. In industrialized countries, severe deficiency is rare, but subclinical deficiency is much more common than people think, especially in those who absorb fats poorly.
The causes of vitamin A deficiency
Vitamin A is a fat-soluble vitamin. This word changes everything. Fat-soluble means it needs dietary fats to be absorbed in the intestine, bile salts to be emulsified, a functioning liver to be stored, and specific transport proteins to circulate in the blood. Each link in this chain can break, and when it does, deficiency sets in insidiously.
The first cause, the most common in developed countries, is fat malabsorption. Anything that reduces bile secretion or impairs lipid absorption directly compromises vitamin A absorption. Biliary insufficiency, whether related to cholecystectomy (as in Nathalie’s case), gallstones, hepatic steatosis, or an overburdened liver, drastically reduces the capacity to emulsify fats in the duodenum. Without bile, micelles don’t form, and vitamin A remains in the intestinal lumen instead of being absorbed by enterocytes2. Celiac disease, Crohn’s disease, cystic fibrosis, chronic pancreatitis, and short bowel syndrome are all pathologies that impair this absorption. It’s exactly the same mechanism that affects vitamin D and vitamin E absorption, because all fat-soluble vitamins share the same absorption pathways.
The second cause is insufficient dietary intake. We’re talking here about two distinct forms of vitamin A. Preformed retinol (or active vitamin A) is found exclusively in animal products: liver, cod liver oil, butter, egg yolk, fatty cheeses. It’s the form directly usable by the body. Beta-carotene (or provitamin A) is found in colored vegetables: carrot, sweet potato, spinach, mango, apricot. But beta-carotene isn’t vitamin A. It’s a precursor that must be converted to retinol by the enzyme BCO1 (beta-carotene 15,15’-monooxygenase) in the enterocyte. And this conversion is very inefficient: on average, 12 micrograms of dietary beta-carotene are needed to produce 1 microgram of retinol3. That’s a ratio of 12 to 1. For some people, it’s even worse.
The third cause is a factor that conventional medicine almost completely ignores: genetic polymorphisms in the BCO1 enzyme. Approximately 45 percent of the European population carries one or two variants of the BCO1 gene that reduce beta-carotene to retinol conversion by 30 to 70 percent. These people are what researchers call “low converters.” They can eat kilos of carrots without ever getting enough active retinol. The clinical sign I look for in consultation: skin yellowing in the palms of the hands and soles of the feet (carotenodermia) despite normal carrot consumption. This yellowing, often confused with hepatic jaundice, actually testifies to an accumulation of unconverted beta-carotene. It’s an indirect sign of BCO1 polymorphism. In these patients, only preformed retinol of animal origin allows their needs to be met.
The fourth cause is hypothyroidism. The thyroid hormone T3 is necessary for BCO1 gene expression. When the thyroid functions slowly, beta-carotene to retinol conversion is compromised. Dr. Hertoghe emphasizes in The Hormone Handbook that hypothyroidism is an underdiagnosed cause of vitamin A deficiency, and that carotenodermia (yellow skin) is a classic sign of hypothyroidism in his clinical practice. The circle is vicious, because vitamin A itself is a cofactor of the T3 receptor, as we’ll see later.
The symptoms of deficiency
The eyes are the first organ to suffer from vitamin A deficiency, and this makes sense when you understand the biochemistry. Vitamin A, in its retinal form (aldehyde of retinol), is the chromophore of rhodopsin, the photosensitive pigment of retinal rods. Rods are the cells responsible for vision in low light, scotopic vision. When a photon of light strikes rhodopsin, 11-cis-retinal transforms into all-trans-retinal, triggering the nerve signal. Then the all-trans-retinal must be recycled into 11-cis-retinal for the cycle to begin again. Without sufficient vitamin A intake, this cycle slows, and night vision degrades.
Hemeralopia (from Greek hemera, day, and ops, eye), or night blindness, is the first clinical sign of vitamin A deficiency. It’s the sign that Nathalie had presented for a year without anyone making the connection. Hemeralopia is reversible if treated promptly. But if deficiency continues, it evolves to xerophthalmia: the conjunctiva and cornea dry out, losing their brightness. Whitish spots appear on the conjunctiva, Bitot’s spots, consisting of accumulated keratin and saprophytic bacteria. Without treatment, the cornea ulcerates (keratomalacia), and blindness becomes irreversible. The WHO estimates that 250,000 to 500,000 children become blind each year due to vitamin A deficiency worldwide1.
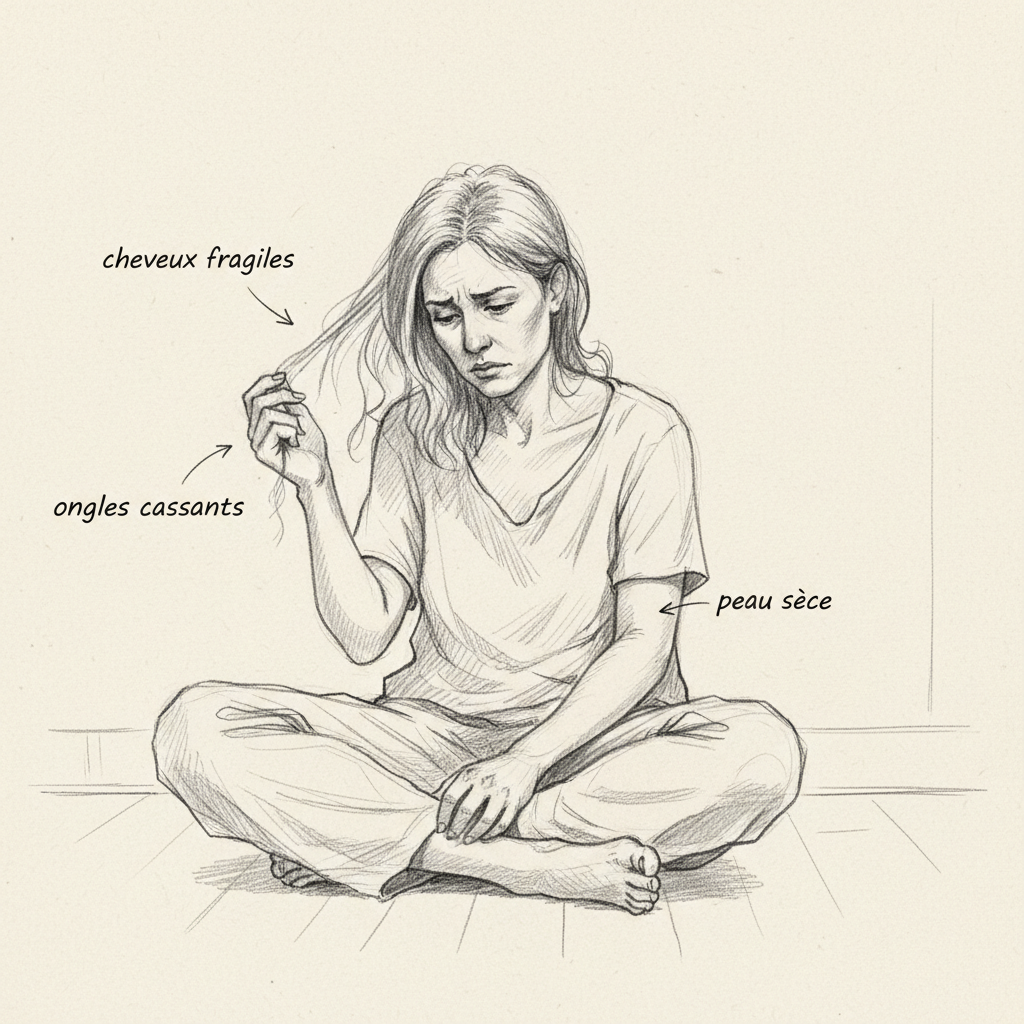
The skin is the second mirror of deficiency. Vitamin A, in its retinoic acid form, regulates keratinocyte differentiation, the cells that renew the epidermis. When it’s lacking, keratinization becomes chaotic: skin cells multiply without maturing properly, and they accumulate excess keratin. This is follicular hyperkeratosis: the skin becomes dry, rough, covered with small horny papules around hair follicles, especially on the arms, thighs, and buttocks. It’s called “permanent goosebumps” skin. Mucous membranes are also affected: dry mouth, irritated throat, dry vaginal mucosa, fragile bronchial mucosa. Retinoic acid is the master regulator of epithelial differentiation throughout the body. Without it, each mucous surface loses its ability to produce protective mucus and renew itself properly.
Immunity is the third pillar that vitamin A deficiency shakes. The immunological role of vitamin A is dual and fundamental. First axis: vitamin A is indispensable for the differentiation of naive T lymphocytes into regulatory T lymphocytes (Treg) in the intestine. Dendritic cells of the intestinal mucosa produce retinoic acid that guides T lymphocyte maturation toward a tolerogenic profile4. Without vitamin A, Tregs don’t form properly, and intestinal immunity loses its ability to distinguish self from non-self. This mechanism directly connects to the issue of autoimmunity and intestinal permeability. Second axis: vitamin A stimulates the production of secretory IgA, the antibodies that line mucous membranes (intestinal, respiratory, urogenital) and constitute the first line of defense against pathogens. Vitamin A deficiency reduces IgA production by 40 to 60 percent according to studies5, which explains why respiratory and digestive infections are so frequent in the deficient. Nathalie, with her recurrent otitis and bronchitis, perfectly illustrated this mechanism.
Vitamin A also plays a role in fertility. Retinoic acid is necessary for spermatogenesis in men and normal embryonic development in women. Sertoli cells in the testes and the uterine endometrium are tissues highly dependent on retinoic acid for their renewal. In women of childbearing age, vitamin A deficiency can contribute to implantation disorders and early miscarriages, although this link needs further study. Preconception should include assessment of vitamin A status.
The micronutrients essential to vitamin A
Vitamin A never works alone. It’s part of a network of cofactors whose deficiency compromises the functioning of all the others. The most important of these cofactors is zinc. Zinc is indispensable at two crucial levels. First, it’s necessary for the synthesis of RBP (Retinol Binding Protein), the transport protein that carries retinol from the liver to target tissues. Without zinc, retinol remains sequestered in the liver and cannot reach the cells that need it6. This is a cause of functional vitamin A deficiency even when hepatic reserves are normal. Second, zinc is a cofactor of retinol dehydrogenase, the enzyme that converts retinol to retinal in the retina. Without zinc, the visual cycle of rhodopsin is compromised, and hemeralopia develops even with adequate vitamin A reserves. Dr. Curtay, in Nutritherapy, emphasizes the zinc-vitamin A interdependence: correcting one without correcting the other is often insufficient.
Vitamin D is an essential partner. The nuclear receptors of vitamin A (RAR and RXR) and vitamin D (VDR) function as heterodimers: the RXR (retinoic acid X receptor) is the mandatory partner of the VDR. In other words, the vitamin D receptor cannot function without a vitamin A receptor to form the active transcriptional complex. Vitamin A deficiency can therefore compromise vitamin D action, even if the 25-OH-D3 level is optimal. This is a biochemical detail that very few practitioners know.
Dietary fats are an indispensable absorption cofactor. Since vitamin A is fat-soluble, taking it during a meal containing lipids (butter, olive oil, avocado, nuts) multiplies its absorption by two to three times. Very low-fat diets, still recommended by some doctors to reduce cholesterol, are an iatrogenic cause of fat-soluble vitamin malabsorption. Bile is the second absorption vector: it emulsifies dietary fats into micelles, and it’s within these micelles that vitamin A is transported to enterocytes. People who have undergone cholecystectomy or suffer from biliary insufficiency are therefore at increased risk.
Iron participates in vitamin A metabolism. Vitamin A improves iron mobilization from ferritin reserves and increases its incorporation into hemoglobin. In return, iron is necessary for certain enzymes in retinoid metabolism. A study by Zimmermann showed that combined iron-vitamin A supplementation is more effective than either alone in correcting anemia in deficient populations7. This is a concrete example of micronutrient synergy.
Dietary sources
The liver is by far the most concentrated source of preformed vitamin A. Cod liver contains approximately 30,000 micrograms of retinol per 100 grams, more than thirty times the recommended daily intake. Cod liver oil contains 18,000 micrograms per 100 grams. Beef liver provides 9,442 micrograms per 100 grams. Poultry liver (chicken, turkey) contains between 3,300 and 4,000 micrograms per 100 grams. These concentrations explain why weekly liver consumption far exceeds needs, and why traditional peoples who regularly consumed organ meats never suffered from vitamin A deficiency. Weston Price, in his studies on indigenous peoples, observed that the healthiest populations consistently consumed foods rich in fat-soluble vitamins, with liver taking first place.
Raw butter (unpasteurized when available) provides 750 micrograms per 100 grams. Egg yolk contains approximately 380 micrograms per egg (or 1,400 micrograms per 100 grams of pure yolk). Egg yolk has the advantage of simultaneously providing vitamin A, vitamin D, choline, and phospholipids in a natural ratio that industry has never been able to reproduce. Fatty cheeses (Beaufort, Comté, Roquefort) provide between 250 and 350 micrograms per 100 grams. Fatty fish (eel, mackerel, bluefin tuna) contain between 150 and 900 micrograms per 100 grams depending on the species.
On the plant side, sources are in beta-carotene, with the 12:1 conversion ratio to keep in mind. Cooked orange sweet potato is the star of carotenoids with approximately 1,000 micrograms of retinol equivalent (RE) per 100 grams. Cooked carrot contains 850 micrograms RE per 100 grams. Cooked spinach provides 470 micrograms RE per 100 grams. Mango contains 54 micrograms RE per 100 grams. Pumpkin and butternut squash provide between 250 and 400 micrograms RE per 100 grams. Fresh parsley contains 420 micrograms RE per 100 grams.
One essential point: gentle cooking and the addition of fat significantly improve beta-carotene bioavailability. Beta-carotene is trapped in the cell walls of vegetables, and cooking breaks these walls. A cooked carrot with a drizzle of olive oil provides three to five times more bioavailable beta-carotene than a raw carrot. This is one of the cases where gentle cooking is preferable to raw for nutrient assimilation.
The antagonists of vitamin A
Alcohol is the first antagonist. Ethanol and vitamin A share the same hepatic detoxification pathways, particularly the enzymes alcohol dehydrogenase (ADH) and aldehyde dehydrogenase (ALDH). When the liver is mobilized to metabolize ethanol, it can no longer properly convert retinol into its active metabolites. Chronic alcohol reduces hepatic vitamin A reserves and simultaneously increases the production of toxic retinol metabolites. This is a double penalty: less functional vitamin A and more harmful metabolites. Dr. Mouton, in Digestive Ecology, classifies chronic alcohol as the most powerful factor for vitamin A depletion in Western countries.
Proton pump inhibitors (PPIs) constitute the second antagonist. PPIs, massively prescribed for acid reflux, reduce the gastric acidity necessary to release protein-bound vitamin A from food. Furthermore, by reducing duodenal acidity, they impair the secretion of cholecystokinin (CCK) that stimulates gallbladder contraction and bile release. Less bile means fewer micelles, and fewer micelles means less vitamin A absorption. This is the mechanism found in all iatrogenic fat-soluble vitamin deficiencies.
Cholestyramine is a bile acid sequestrant used to reduce cholesterol. It traps bile salts in the intestine and prevents their reabsorption. Direct consequence: dietary fats and fat-soluble vitamins (A, D, E, K) are no longer emulsified and pass through the digestive tract without being absorbed. Long-term patients on cholestyramine develop fat-soluble vitamin deficiencies within six to twelve months of starting treatment if no supplementation is implemented.
Orlistat (Xenical, Alli), a pancreatic lipase inhibitor prescribed for weight loss, blocks the absorption of thirty percent of dietary fats. Unabsorbed fats carry fat-soluble vitamins with them. Orlistat reduces vitamin A absorption by thirty percent on average, and manufacturers explicitly recommend fat-soluble vitamin supplementation during treatment. How many patients on orlistat never received this information.
Mineral oil, used as a laxative or cooking oil in certain weight-loss practices, is a trap for fat-soluble vitamins. Not being absorbed by the intestine, it passes through the digestive tract dissolving vitamins A, D, E, and K as it goes, carrying them away in the stool. It’s a mechanically redoubtable antagonist.
Fat malabsorption in the broad sense (exocrine pancreatic insufficiency, cystic fibrosis, intestinal resection, Whipple’s disease) constitutes the last major antagonist. Any situation that reduces the body’s ability to digest and absorb dietary lipids cascadingly compromises the absorption of all four fat-soluble vitamins.
The forgotten causes of deficiency
The link between vitamin A and the thyroid is a subject that few practitioners master, and which would deserve its own chapter in endocrinology textbooks. Retinoic acid is a cofactor of the T3 receptor (the nuclear receptor TR). The T3 receptor forms a heterodimer with RXR, the retinoic acid receptor, exactly as the vitamin D VDR does. Without vitamin A, the T3 signal is attenuated at the genomic level, even if blood T3 levels are normal. This is a cause of tissue hypothyroidism invisible to standard analysis. In the other direction, hypothyroidism reduces BCO1 activity and compromises beta-carotene to retinol conversion. The thyroid-vitamin A vicious circle is a clinical trap that Dr. Hertoghe describes with precision: correcting the thyroid improves vitamin A status, and correcting vitamin A improves tissue response to T3.
The gut microbiota plays an underrecognized role in vitamin A metabolism. Recent studies show that certain intestinal bacterial strains (notably Lactobacillus and Bifidobacterium) influence BCO1 expression in enterocytes and modulate retinol absorption8. Severe intestinal dysbiosis can impair vitamin A metabolism independently of dietary intake. This is yet another illustration of the central role of the intestine in nutrition, which naturopathy has been asserting for decades: the first pillar of health is the quality of your intestine.
Chronic oxidative stress consumes vitamin A. Retinol and beta-carotene are antioxidants, and in situations of significant oxidative stress (smoking, pollution, chronic inflammation, intense sport without recovery), they’re consumed to neutralize free radicals, which reduces the amount available for their physiological functions (vision, immunity, cell differentiation). This is the same “accelerated consumption” mechanism observed with zinc in stress situations.
Type 2 diabetes and insulin resistance impair hepatic vitamin A metabolism. Steatotic liver (fatty liver), extremely common in diabetics and overweight people, stores retinol less efficiently and produces less RBP. Hepatic stellate cells (Ito cells), which normally store eighty percent of the body’s vitamin A reserves, transform into myofibroblasts in case of steatosis and fibrosis, thus losing their storage capacity. This is a mechanism that Mouton documents in Digestive Ecology: fatty liver is a liver that no longer stores fat-soluble vitamins properly.
Drug interactions are an iatrogenic cause too often neglected. Beyond PPIs, cholestyramine, and orlistat already mentioned, synthetic retinoids (isotretinoin, acitretin) prescribed in dermatology saturate RAR receptors and can paradoxically induce signs of functional natural vitamin A deficiency while generating an excess of synthetic retinoids. Metformin, while not directly interfering with vitamin A, can reduce B12 absorption on which folate metabolism depends, and folates indirectly participate in retinoid recycling. These cascading interactions illustrate why naturopathy always looks at the terrain as a whole rather than an isolated nutrient.
Dietary supplements
Vitamin A supplementation comes in two main forms, and the choice between them depends on the clinical context.
Retinyl palmitate (or retinol palmitate) is the most common preformed form in dietary supplements. It’s a retinol ester that’s hydrolyzed in the intestine to release free retinol, directly absorbable. The advantage of retinyl palmitate is that it doesn’t depend on the BCO1 enzyme to be converted: it’s active vitamin A. The usual dosage in adults is 800 to 1,500 micrograms (2,600 to 5,000 IU) per day. The maximum tolerated dose in non-pregnant adults is 3,000 micrograms (10,000 IU) per day according to the EFSA. Beyond that, the risk of hypervitaminosis A (hepatotoxicity, headaches, nausea, skin peeling) increases, especially with chronic use. Always take retinyl palmitate with the fattiest meal of the day to optimize absorption.
Natural beta-carotene is the plant-based alternative. Natural beta-carotene, extracted from the alga Dunaliella salina or red palm oil, contains a mixture of carotenoids (alpha-carotene, beta-carotene, lutein, zeaxanthin) that act synergistically. It fundamentally differs from synthetic beta-carotene (pure all-trans-beta-carotene) used in some low-cost supplements. The ATBC study (Alpha-Tocopherol, Beta-Carotene Cancer Prevention Study) and the CARET study showed that high-dose synthetic beta-carotene (20 to 30 milligrams per day) increased lung cancer risk in smokers, probably by a paradoxical pro-oxidant effect at high doses. Natural beta-carotene at physiological doses (6 to 15 milligrams per day) has never shown this risk. The distinction is crucial. The great advantage of beta-carotene is its safety: the body only converts to retinol what it needs, and excess is stored without toxicity (it colors the skin yellow-orange, that’s all). This is why natural beta-carotene is the preferred form during pregnancy, where the teratogenic risk of high-dose preformed retinol is well documented. The maximum dose of preformed retinol during pregnancy is 3,000 micrograms (10,000 IU) per day, and in practice I recommend not exceeding 1,500 micrograms of retinol in the first trimester, favoring natural beta-carotene for supplementation.
For people with known or suspected BCO1 polymorphism (carotenodermia, yellow skin on palms despite normal carotenoid intake), preformed retinol supplementation is preferable to beta-carotene, since the conversion pathway is deficient. Zinc bisglycinate (15 to 30 milligrams per day) should systematically accompany vitamin A supplementation to ensure RBP synthesis and retinol transport to target tissues.
In summary, the protocol I use in practice to correct vitamin A deficiency rests on three pillars: first, restore digestive function (biliary support through artichoke, black radish, or desmodium, correction of hypochlorhydria if necessary, intestinal repair if dysbiosis or permeability). Second, provide vitamin A in the form adapted to the patient’s profile (retinyl palmitate if documented deficiency, natural beta-carotene if prevention or pregnancy). Third, correct associated cofactors: zinc, vitamin D, quality fats, and thyroid evaluation if conversion appears deficient.
Want to assess your vitamin A deficiency risk? Take the vitamin A deficiency questionnaire in 2 minutes. And if you also suspect an associated thyroid problem, the Claeys test completes the assessment.
Nathalie, my patient from the beginning, followed this protocol for four months. Daily biliary support (artichoke and desmodium, because her liver without a gallbladder needed permanent assistance), retinyl palmitate 1,500 micrograms per day at dinner, zinc bisglycinate 30 milligrams in the evening, and vitamin D3 4,000 IU per day (her level was 28 ng/mL). After six weeks, her skin had regained elasticity. After two months, she had no more otitis. And after three months, she told me the sentence I was waiting for: “I drive at night again without problems.” Her ophthalmologist, when she told him about it, didn’t make the vitamin A connection. He said her vision had “improved spontaneously.” Spontaneously. It’s a word medicine uses when it hasn’t looked for the cause.
For vitamin A and zinc supplementation, Sunday Natural offers pharmaceutical-grade retinyl palmitate and natural beta-carotene, precisely dosed (-10% with code FRANCOIS10). Find all my partnerships with exclusive promo codes.
For further reading
- Vitamin B2 (riboflavin): Your mitochondria won’t run without it
- Vitamin B8 (biotin): Hair, skin, and blood sugar in one molecule
- Vitamin C: Immunity, collagen, and antioxidant shield
- Zinc: Why you’re probably deficient (and what to do about it)
Sources
- Curtay, Jean-Paul. Nutritherapy. Marco Pietteur, 2016.
- Hertoghe, Thierry. The Hormone Handbook. 2nd ed. Luxembourg: International Medical Books, 2012.
- Mouton, Georges. Digestive Ecology. Marco Pietteur, 2004.
- Sommer, Alfred. Vitamin A Deficiency and Its Consequences. 3rd ed. WHO, 1995.
“Food is your first medicine. Unless, of course, your body knows how to absorb it.” Pierre-Valentin Marchesseau
Scientific references
Footnotes
-
Sommer, Alfred. “Vitamin A Deficiency and Clinical Disease: An Historical Overview.” Journal of Nutrition 138, no. 10 (2008): 1835-1839. PMID: 18806089. ↩ ↩2
-
Blomhoff, Rune, et al. “Transport and Storage of Vitamin A.” Science 250, no. 4979 (1990): 399-404. PMID: 2218545. ↩
-
Tang, Guangwen. “Bioconversion of Dietary Provitamin A Carotenoids to Vitamin A in Humans.” American Journal of Clinical Nutrition 91, no. 5 (2010): 1468S-1473S. PMID: 20200262. ↩
-
Iwata, Makoto, et al. “Retinoic Acid Imprints Gut-Homing Specificity on T Cells.” Immunity 21, no. 4 (2004): 527-538. PMID: 15485630. ↩
-
Villamor, Eduardo, and Wafaie W. Fawzi. “Effects of Vitamin A Supplementation on Immune Responses and Correlation with Clinical Outcomes.” Clinical Microbiology Reviews 18, no. 3 (2005): 446-464. PMID: 16020684. ↩
-
Christian, Parul, and Keith P. West. “Interactions between Zinc and Vitamin A: An Update.” American Journal of Clinical Nutrition 68, no. 2 (1998): 435S-441S. PMID: 9701158. ↩
-
Zimmermann, Michael B., et al. “Dual Fortification of Salt with Iodine and Microencapsulated Iron: A Randomized, Double-Blind, Controlled Trial in Moroccan Schoolchildren.” American Journal of Clinical Nutrition 80, no. 4 (2004): 952-959. PMID: 15447905. ↩
-
Lv, Zhishuo, et al. “Vitamin A and the Gut Microbiota: A Reciprocal Interaction.” Critical Reviews in Food Science and Nutrition 62, no. 11 (2022): 3029-3042. PMID: 33412968. ↩





Laisser un commentaire
Sois le premier à commenter cet article.