Your doctor says “your vitamin D is within normal range.” You look at the results: 31 ng/mL. The lab reference range says “normal above 30.” You file the paper away, reassured. Except you have no reason to be reassured. Because the threshold of 30 ng/mL is the threshold below which you risk rickets or osteomalacia. It’s the threshold of bone disease, not the threshold of health. Scientific research from the past twenty years shows that an optimal level falls between 50 and 80 ng/mL1. Between 31 and 50, your bones, your immunity, your mood and your cancer protection suffer in silence. And nobody tells you.
“The ESVITAF survey shows that more than 90% of French women and men have insufficient vitamin D intake. It’s the most massive deficit in our population, and it’s also the one we correct the least well, because we confuse lab standards with health standards.” Dr Anne Lucas, PharmD, DU MAPS 2020, course #33
When I heard this sentence in the lecture hall, I thought of all the patients I’d seen in consultation with a vitamin D level of 32, 34, 37 ng/mL and whom their doctor had told “it’s fine.” It’s not fine. It’s the bare minimum. And the difference between bare minimum and optimal is the difference between surviving and living in full health.
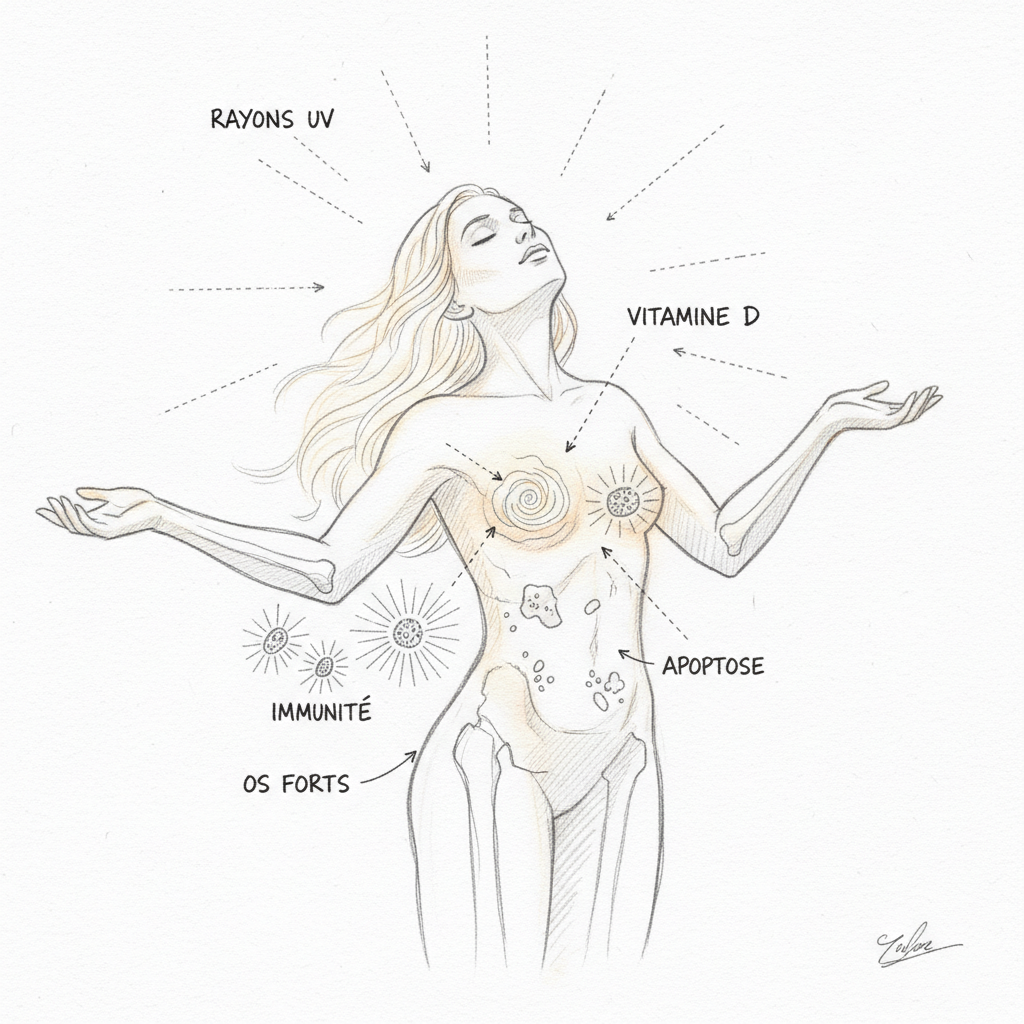
The first thing to understand, and it’s the most important, is that vitamin D is not a vitamin. It’s a hormone. A real hormone, with a dedicated nuclear receptor (the VDR, Vitamin D Receptor) that directly regulates the expression of more than 200 genes2. The confusion comes from history: when it was discovered in the early twentieth century, it was classified as a vitamin because it seemed to come exclusively from food. We now know that 80 to 90% of vitamin D is synthesized by the skin under the effect of UVB rays. It doesn’t come from your plate. It comes from the sun. That’s why it’s called the sun hormone. It’s exactly the same conceptual error we make with the thyroid: we reduce vitamin D to its bones, just as we reduce the thyroid to its TSH. In both cases, we miss the essential.
Want to assess your risk of deficiency? Take the vitamin D deficiency questionnaire in 2 minutes.
Causes of vitamin D deficiency
Vitamin D deficiency is the most widespread deficiency in temperate countries. If you use lab standards (30 ng/mL), about 40% of the French population is deficient. If you use the 50 ng/mL threshold, it’s 80 to 90% of the population that is in functional insufficiency. And in winter, north of the 42nd parallel (which includes all of mainland France except the far south), the situation is even worse because skin synthesis is virtually zero from November to February. Dr Lucas asked in class: “Why haven’t lab standards been updated despite twenty years of research?” The answer is systemic. If we raised the normal threshold to 50 ng/mL, we would have to massively supplement the population, which would have a cost for health insurance. It’s an economic choice disguised as a scientific choice.
The primary cause is latitude. Skin synthesis only occurs with UVB rays of wavelength 290-315 nm. These UVBs reach the earth’s surface in sufficient quantity only between April and October in France, and only between approximately 11am and 3pm. During the six winter months, the sun is too low on the horizon for UVBs to pass through the atmosphere. All of mainland France is located above the 42nd parallel, which means that for six months, vitamin D synthesis is biologically impossible through skin exposure. Nordic countries are obviously the most affected, and it’s in these countries that we observe the highest rates of multiple sclerosis, type 1 diabetes, and winter depression.
Indoor lifestyle is the second cause. We spend an average of 90% of our time indoors (office, transportation, home). UVBs don’t penetrate glass. Working behind a sunny window doesn’t produce a single nanogram of vitamin D. It’s a reality that many people don’t know. And even when they go outside, it’s often early morning or late afternoon, when the solar angle is too low to allow skin synthesis.
Skin color is a determining factor. Melanin, the pigment that gives skin its color, absorbs UVBs. It’s a natural sunscreen. Dark and Black skin requires three to five times more exposure time to synthesize the same amount of vitamin D as light skin1. Populations of African, Caribbean, or North African origin living in mainland France are almost systematically deficient. It’s massive epidemiological data yet rarely integrated into medical recommendations.
Age greatly worsens the situation. The skin of a 70-year-old synthesizes four times less vitamin D than that of a 20-year-old, because the concentration of 7-dehydrocholesterol (the skin precursor) decreases with aging. The kidney also converts 25-OH-D3 less efficiently to active form. The link with sarcopenia (muscle mass loss), fall risk, and osteoporosis is well documented, and it’s a major issue in aging prevention.
Obesity is an underrecognized but powerful factor. Vitamin D, being fat-soluble, is sequestered in adipose tissue and becomes less bioavailable. Obese individuals have significantly lower vitamin D levels than people of normal weight, and require supplementation doses 1.5 to 2 times higher to reach the same blood level. It’s a biochemical trap: the more body fat you have, the more your vitamin D is trapped in adipose tissue instead of circulating in the blood.
Pregnant women have increased needs, and the mother’s vitamin D status determines that of the newborn. Maternal deficiency is associated with increased risk of pre-eclampsia, gestational diabetes, and neonatal rickets. Systematic supplementation during pregnancy is a bare minimum that naturopathy has defended for years.
Symptoms of deficiency
The VDR receptor is a transcription factor. When calcitriol (active form) binds to it, it forms a complex with another receptor (the RXR), and this complex binds to DNA at specific sequences called VDREs (Vitamin D Response Elements). This process activates or suppresses the transcription of more than 200 genes2. The VDR is present in virtually all tissues in the body: intestine, bone, muscle, brain (hippocampus), immune system (T and B lymphocytes, macrophages, dendritic cells), pancreas, breast, prostate, colon3. When you lack vitamin D, it’s not just your bones that suffer. Every tissue that has a VDR suffers. And they’re everywhere.
Rickets and osteoporosis are the classic symptoms. Vitamin D stimulates the production of calbindin in the enterocyte, a transport protein that allows active absorption of dietary calcium. Without vitamin D, you absorb only 10 to 15% of ingested calcium. With an optimal level, you absorb 30 to 40%. It’s a difference that over years makes the difference between solid bones and osteoporosis. In children, it’s rickets. In adults, it’s osteomalacia then osteoporosis. But these bone diseases are only the tip of the iceberg.
Recurrent infections are a cardinal sign. Vitamin D stimulates innate immunity by increasing the production of cathelicidin (LL-37) and defensins, antimicrobial peptides that destroy bacteria, viruses, and fungi4. That’s why winter infections explode when vitamin D levels drop. Every winter, the same pattern repeats: vitamin D levels collapse, colds, flus, bronchitis multiply, and nobody makes the connection with the sun that disappeared six months earlier. In consultation, when a patient has a string of ENT infections since October, I systematically measure 25-OH-D3. In nine out of ten cases, the level is below 30 ng/mL.
Autoimmune diseases constitute the most serious and most underestimated manifestation. Vitamin D modulates adaptive immunity by restraining Th1 and Th17 responses (pro-inflammatory and autoimmune) while promoting Treg responses (regulatory, tolerogenic). In other words: vitamin D prevents your immune system from going haywire against your own tissues. Hashimoto, the most common autoimmune thyroiditis, is associated in numerous studies with low vitamin D levels. A meta-analysis published in Thyroid shows that Hashimoto patients have significantly lower 25-OH-D3 levels than healthy subjects5. Multiple sclerosis presents the same profile: its prevalence increases with latitude, and intervention studies show that vitamin D supplementation significantly reduces the number of relapses. Type 1 diabetes, an autoimmune disease that destroys pancreatic beta cells, is also associated with vitamin D deficiency: a Finnish study showed that supplementation in the first year of life reduced the risk by 80% over a thirty-year follow-up. In consultation, when a patient presents with an autoimmune pathology (endometriosis, rheumatoid arthritis, lupus, Crohn’s disease), 25-OH-D3 measurement is part of my systematic evaluation.
Depression and cognitive decline are frequent neuropsychiatric symptoms. The VDR is strongly expressed in the hippocampus, the brain region essential for memory and mood regulation. Studies show that low vitamin D levels are associated with increased risk of depression, cognitive decline, and Alzheimer’s6. Vitamin D stimulates the production of BDNF (Brain-Derived Neurotrophic Factor), the same growth factor that physical exercise stimulates to protect neurons. It’s a link I explore in detail in the article on brain and cognition.
Cancer is the last major chapter. The VDR is present in breast, prostate, colon, and pancreas cells. When calcitriol binds to the VDR of these cells, it stimulates differentiation (cells mature and specialize instead of proliferating chaotically) and apoptosis (defective cells self-destruct). The meta-analyses by Garland and Garland show that a 25-OH-D3 level above 40 ng/mL is associated with a 50% reduction in colorectal cancer risk compared to a level below 20 ng/mL7. Similar results have been observed for breast, prostate, and pancreatic cancers. The mechanism passes through four complementary anticancer pathways: cell differentiation, proliferation braking, apoptosis of precancerous cells, and inhibition of tumor angiogenesis. They are all dependent on vitamin D level.
Essential micronutrients for vitamin D
Vitamin D alone is not enough. To understand why, we need to go back to the synthesis and activation chain. It all starts in the skin. UVB rays convert 7-dehydrocholesterol into pre-vitamin D3, which transforms into cholecalciferol (vitamin D3) under the effect of body heat. Cholecalciferol travels in the blood, bound to DBP (Vitamin D Binding Protein), to the liver where the enzyme 25-hydroxylase (CYP2R1) converts it into 25-OH-D3 (calcidiol), the form measured in the blood. 25-OH-D3 then travels to the kidney where 1-alpha-hydroxylase (CYP27B1) converts it into 1,25-(OH)2-D3 (calcitriol), the active form that binds to the VDR. Each step in this chain depends on precise cofactors.
Magnesium is the most critical cofactor. Both hydroxylations (hepatic and renal) require magnesium as an enzymatic cofactor8. A magnesium deficiency blocks vitamin D activation, even if you’re taking massive doses. That’s why many patients who take vitamin D don’t see their level rise sufficiently: they lack magnesium to activate it. The cycle is vicious: magnesium deficiency prevents vitamin D activation, and vitamin D deficiency worsens renal magnesium loss. Correcting both together is essential. Magnesium bisglycinate, at 300 to 400 mg per day with taurine and B6, is the form I systematically recommend in association with vitamin D.
Vitamin K2-MK7 is the second essential cofactor. Its role is to direct calcium to the right destinations. Vitamin D increases intestinal calcium absorption, but it doesn’t control where this calcium is deposited. Without K2, calcium can deposit in arteries (vascular calcifications), kidneys (kidney stones), or joints (calcifying arthropathy) instead of in bones. It’s the calcium paradox: studies show that calcium supplementation alone, without K2, increases cardiovascular risk while reducing fracture risk. K2-MK7 resolves this paradox by activating two calcium-dependent proteins: osteocalcin (which fixes calcium in bone) and MGP (Matrix Gla Protein, which prevents arterial calcification). The vitamin D + K2 duo is the physiological pair: D brings calcium in, K2 directs it to the right place. The usual dose is 100 to 200 micrograms of K2-MK7 per day.
Zinc is the third cofactor not to overlook. Zinc is a cofactor of the VDR itself. Without zinc, the vitamin D receptor functions less well, which reduces the effectiveness of active vitamin D even when the blood level is correct. Zinc is also involved in T4 to T3 conversion at the thyroid level, and thyroid and vitamin D share many metabolic interactions. A combined zinc-vitamin D deficiency is extremely common and creates a terrain conducive to autoimmune diseases.
Food sources
Food is structurally incapable of meeting our vitamin D needs. Maximum food intake is around 200 to 400 IU per day, while skin synthesis produces between 10,000 and 20,000 IU in 20 minutes of full-body exposure. This order of magnitude shows how much vitamin D is above all a solar hormone, not a food nutrient. That said, certain food sources usefully contribute to vitamin D status, especially in winter when skin synthesis is zero.
Cod liver oil is the most concentrated food source, with approximately 1360 IU per tablespoon. Our grandparents took it every winter, and it was a remarkably effective public health measure. It provides in addition vitamin A and omega-3, making it a complete functional food. Fatty fish (wild salmon, mackerel, sardine, herring) provide between 400 and 600 IU per 100 grams. Wild salmon contains about four times more vitamin D than farmed salmon, a difference explained by natural plankton diet.
Egg yolk provides approximately 40 IU per unit, which is modest but cumulative if you consume them regularly. Eggs from outdoor-raised hens contain more vitamin D than cage-raised eggs, because hens exposed to sun synthesize more vitamin D3 that ends up in the yolk. Butter and fatty cheeses provide low but non-negligible quantities. Calf liver is an interesting source (approximately 50 IU per 100 grams) in addition to its iron, zinc, and vitamin A content.
Mushrooms are the only plant source of vitamin D, but as D2 (ergocalciferol), less effective than D3. Button mushrooms exposed to UV contain approximately 400 IU per 100 grams, but their content is highly variable depending on growing conditions. It’s a backup source for vegans, nothing more.
Sun exposure remains the physiological source par excellence. From April to October, expose arms and legs to the sun without sunscreen for 15 to 30 minutes per day (light skin) or 30 to 60 minutes (dark skin), between 11am and 3pm. This isn’t cosmetic advice, it’s a health action. Light therapy doesn’t compensate for vitamin D deficiency (light therapy lamps emit visible light, not UVBs). Only supplementation works from November to March at our latitudes.
Vitamin D antagonists
Sunscreens are the first antagonist, and the most paradoxical. SPF 30 sunscreen blocks 97% of UVBs, which reduces skin synthesis of vitamin D in the same proportions. Systematic sunscreen use on all exposed skin, promoted by melanoma prevention campaigns, had a massive collateral effect on the population’s vitamin D status. The reasonable compromise is to expose arms and legs without sunscreen for 15 to 30 minutes before applying sunscreen to at-risk areas (face, décolletage, shoulders).
Obesity acts as a vitamin D trap. Adipose tissue sequesters fat-soluble vitamin D and makes it less bioavailable. It’s a storage mechanism that becomes pathological when excess body fat is present: vitamin D is trapped in adipocytes instead of circulating in the blood. Obese people require 1.5 to 2 times higher doses to reach the same blood level.
Statins can interfere with vitamin D synthesis. 7-dehydrocholesterol, the skin precursor of vitamin D, is a cholesterol derivative. Statins inhibit HMG-CoA reductase upstream of the cholesterol synthesis pathway, which can theoretically reduce the availability of 7-dehydrocholesterol. The clinical impact remains debated, but it’s a factor to consider in patients on statins with low vitamin D levels.
Corticosteroids (cortisone, prednisone) accelerate vitamin D catabolism and reduce intestinal calcium absorption. Patients on long-term corticotherapy are systematically at risk of vitamin D deficiency and osteoporosis. Supplementation is essential in this context.
Anticonvulsants (phenytoin, carbamazepine, phenobarbital) induce hepatic enzymes of cytochrome P450 that catabolize 25-OH-D3. Epileptic patients on long-term treatment have significantly lower vitamin D levels and increased fracture risk.
Proton pump inhibitors (PPIs), massively prescribed for acid reflux, reduce dietary vitamin D absorption by decreasing gastric acidity. It’s the same mechanism that disrupts the absorption of magnesium, zinc, and iron. PPIs are micronutrient thieves that medicine prescribes routinely.
Patients suffering from intestinal malabsorption (celiac disease, Crohn’s, severe dysbiosis, intestinal permeability) absorb oral vitamin D poorly, regardless of dose. In these patients, repairing the intestinal barrier must precede or accompany vitamin D supplementation.
Forgotten causes of deficiency
The first forgotten cause, and the most scandalous, is misleading lab standards. Standards set the “normal” threshold for 25-OH-D3 at 30 ng/mL. This threshold was defined to prevent rickets in children and osteomalacia in adults, that is, the most serious bone diseases related to profound deficiency. It’s a survival threshold, not a health threshold. The Endocrine Society recommends 40 to 60 ng/mL. Michael Holick (Boston University) advocates 50 to 80 ng/mL1. Observational studies show that it’s within this range that benefits for immunity, cancer prevention, neurological protection, and muscle health are most pronounced. If we raised the normal threshold to 50 ng/mL, we would have to massively supplement the population. It’s an economic choice disguised as a scientific choice. In naturopathy, we aim for optimal, not minimum.
The second forgotten cause is VDR receptor polymorphism. The VDR, like all nuclear receptors, can present genetic variants (SNP polymorphisms) that modify its efficiency. The most studied polymorphisms are FokI, BsmI, ApaI, and TaqI. Some variants reduce the VDR’s affinity for calcitriol, which means that even with a correct blood level of 25-OH-D3, the biological effect at the cellular level is diminished. These patients need higher blood levels to obtain the same physiological benefit. It’s a possible explanation for those patients who supplement correctly, reach a level of 50 ng/mL, and still present symptoms of deficiency. Nutrigenomics opens exciting perspectives here for personalizing dosages.
The third forgotten cause is the bidirectional link with autoimmunity. We know that vitamin D deficiency promotes autoimmune diseases by removing the brakes on Th1 and Th17 responses. But the reverse is also true. Certain autoimmune diseases, particularly pathologies affecting the intestine (celiac disease, Crohn’s), impair the absorption of oral vitamin D, creating a self-perpetuating vicious circle. The autoimmune process itself consumes vitamin D through macrophage activation (which express 1-alpha-hydroxylase and locally convert 25-OH-D3 to calcitriol for their own immune needs). In Hashimoto patients, autoimmune thyroiditis creates a double handicap: hypothyroidism reduces gastric acid secretion (compromising absorption), and the autoimmune process consumes available vitamin D. This is why autoimmune patients need higher target levels, often above 60 ng/mL.
The fourth forgotten cause is the extra-renal activity of 1-alpha-hydroxylase. Dr Lucas emphasized in class that 1-alpha-hydroxylase is not present only in the kidney. It’s found in macrophages, dendritic cells, and colon and breast cells. These tissues locally produce calcitriol for their own needs, through an autocrine and paracrine mechanism. This means that the active form measured in the blood (1,25-(OH)2-D3) doesn’t reflect local vitamin D activity in tissues. A “normal” 1,25-(OH)2-D3 measurement doesn’t guarantee that your macrophages or intestinal cells receive enough calcitriol. That’s why measuring 25-OH-D3 (reserve form) is the only reliable marker of overall status.
Dietary supplements
Vitamin D exists in two forms: D2 (ergocalciferol), of plant origin (mushrooms, irradiated yeast), and D3 (cholecalciferol), of animal origin (skin, fatty fish, cod liver oil, egg yolk) or produced by lichens for vegan forms. Heaney’s studies show that D3 is approximately three times more effective than D2 in increasing and maintaining blood 25-OH-D3 levels9. D2 has a shorter half-life, lower affinity for DBP (transport protein), and for the VDR. In supplementation, D3 is systematically preferred. D2 is only of interest for strict vegan patients who refuse any animal source, and in that case, the dose must be increased to compensate for the lower effectiveness.
Vitamin D ampoules prescribed in France (such as Uvedose, ZymaD forte) are often D3, which is good. But they’re prescribed in massive spaced-out doses (100,000 IU every three months). This “bolus” strategy is increasingly being questioned. Daily intake of 2000 to 4000 IU maintains much more stable blood levels than a quarterly ampoule that creates a spike followed by progressive decline. The body prefers regularity. In naturopathy, daily supplementation in drops is the standard.
The protocol I use in practice is based on three steps. First step: measure. Request a 25-OH-D3 measurement (not the 1,25-(OH)2-D3 which is the active form and doesn’t reflect reserves). Ideally, measure at the end of winter (March) to have the lowest level, and at the end of summer (September) to have the highest level. The objective is to be between 50 and 80 ng/mL at all times.
Second step: supplement according to starting level. When the level is below 20 ng/mL (severe deficiency), the dosage is 4000 to 5000 IU per day for 3 months, followed by a blood test. Between 20 and 40 ng/mL (insufficiency), 3000 to 4000 IU per day for 3 months. Between 40 and 50 ng/mL (sub-optimal), 2000 to 3000 IU per day in maintenance. Above 50 ng/mL (optimal), 1000 to 2000 IU per day in winter maintenance. Always in D3 (cholecalciferol), always in daily drops, always taken with the most fatty meal of the day (vitamin D is fat-soluble and its absorption is multiplied by 2 to 3 in the presence of lipids). Always combine with K2-MK7 (100 to 200 micrograms per day), magnesium bisglycinate (300 to 400 mg per day with taurine and B6), and zinc (15 to 30 mg per day as a VDR cofactor).
Third step: monitor. A 25-OH-D3 measurement every 3 to 6 months allows dosage adjustment. Vitamin D is fat-soluble and can accumulate in the body, but toxicity signs generally only appear above 150 ng/mL. The safety margin is very wide with doses of 2000 to 4000 IU per day. Patients with sarcoidosis, lymphoma, or primary hyperparathyroidism should be cautious, as these pathologies increase conversion to calcitriol through extra-renal pathways. Patients on anticoagulants (warfarin, fluindione) must inform their doctor of K2-MK7 use, as K2 can interfere with anticoagulation.
The VITAL study (Vitamin D and Omega-3 Trial), published in the New England Journal of Medicine in 2019, showed no significant reduction in overall cancer incidence in the supplemented group. But critics noted that the dose (2000 IU/day) was insufficient to bring all participants to a level above 50 ng/mL, and that the follow-up duration (5 years) was probably too short to observe an effect on carcinogenesis, a process that unfolds over decades. In naturopathy, we don’t wait for proof to be absolute to act. We act when the benefit-risk balance is favorable.
Your 31 ng/mL level is not normal. It’s the minimum. And between the minimum and optimal, there’s a world of health that you deserve to inhabit. Get the test, correct the deficiency, add the cofactors, and never forget that the sun is a free medicine. Dr Anne Lucas reminded us at the DU MAPS: medicine of tomorrow will be preventive or it won’t be. And vitamin D is one of the pillars of this prevention.
If you want personalized support to optimize your vitamin D status, you can book a consultation. For vitamin D3, K2-MK7, and magnesium supplementation, Sunday Natural offers pharmaceutical quality formulations with precise dosages (code FRANCOIS10). Find all my partnerships.
To go further
- Cancer and diet: what micronutrition changes in the equation
- Zinc: why you’re probably deficient (and what to do)
- Alzheimer: the metabolic disease you can prevent 20 years before
- Anemia: understanding root causes and acting naturally
Sources
- Holick, Michael F. “Vitamin D Deficiency.” New England Journal of Medicine 357, no. 3 (2007): 266-281.
- Bouillon, Roger, et al. “Vitamin D and Human Health: Lessons from Vitamin D Receptor Null Mice.” Endocrine Reviews 29, no. 6 (2008): 726-776.
- Manson, JoAnn E., et al. “Vitamin D Supplements and Prevention of Cancer and Cardiovascular Disease.” New England Journal of Medicine 380 (2019): 33-44. (VITAL Study)
- Lucas, Anne. “L’homme métal, l’homme minéral.” Course #33, DU MAPS, Faculty of Pharmacy, 2020.
- Curtay, Jean-Paul. Nutritherapy: scientific foundations and medical practice. Testez Editions, 2016.
- Hertoghe, Thierry. Atlas of hormonal and nutritional medicine. Luxembourg: International Medical Books, 2006.
- Mouton, Georges. Digestive ecology. Marco Pietteur, 2004.
“Health is not the absence of disease. It’s the state in which each cell receives what it needs to function optimally.” Dr Jean-Paul Curtay
Scientific references
Footnotes
-
Holick, Michael F. “Vitamin D Deficiency.” New England Journal of Medicine 357, no. 3 (2007): 266-281. PMID: 17634462. ↩ ↩2 ↩3
-
Bouillon, Roger, et al. “Vitamin D and Human Health: Lessons from Vitamin D Receptor Null Mice.” Endocrine Reviews 29, no. 6 (2008): 726-776. PMID: 18694980. ↩ ↩2
-
Wang, Yingyu, et al. “Where Is the Vitamin D Receptor?” Archives of Biochemistry and Biophysics 523, no. 1 (2012): 123-133. PMID: 22503810. ↩
-
Liu, Philip T., et al. “Toll-Like Receptor Triggering of a Vitamin D-Mediated Human Antimicrobial Response.” Science 311, no. 5768 (2006): 1770-1773. PMID: 16497887. ↩
-
Wang, Jing, et al. “Meta-Analysis of the Association between Vitamin D and Autoimmune Thyroid Disease.” Nutrients 7, no. 4 (2015): 2485-2498. PMID: 25854833. ↩
-
Anglin, Rebecca E.S., et al. “Vitamin D Deficiency and Depression in Adults: Systematic Review and Meta-Analysis.” British Journal of Psychiatry 202, no. 2 (2013): 100-107. PMID: 23377209. ↩
-
Garland, Cedric F., et al. “Vitamin D and Prevention of Colorectal Cancer.” Journal of Steroid Biochemistry and Molecular Biology 97, no. 1-2 (2005): 179-194. PMID: 16236494. ↩
-
Uwitonze, Anne Marie, and Mohammed S. Razzaque. “Role of Magnesium in Vitamin D Activation and Function.” Journal of the American Osteopathic Association 118, no. 3 (2018): 181-189. PMID: 29480918. ↩
-
Heaney, Robert P., et al. “Vitamin D3 Is More Potent than Vitamin D2 in Humans.” Journal of Clinical Endocrinology and Metabolism 96, no. 3 (2011): E447-E452. PMID: 21177785. ↩

Laisser un commentaire
Sois le premier à commenter cet article.