Eight Women for Two Men: The Thyroid-Estrogen Knot That Medicine Never Unties
Eight women for two men. That’s the ratio that should make every endocrinologist, every gynecologist, every doctor prescribing Levothyrox stop and ask themselves why it’s women filling the waiting rooms. Not men. Women. Eight times out of ten.
In five years of thyroid consultations, I’ve seen hundreds of women with the same profile. TSH “within normal range,” free T3 below the functional threshold, chronic fatigue, unexplained weight gain, cold sensitivity, brain fog. And every time, the same unanswered question: why me? Why now? Why since the pill, since pregnancy, since menopause?
The answer comes down to one word that nobody mentions in consultations: estrogens. Not estrogens as vague “female hormones.” Estrogens as direct disruptors of thyroid metabolism, through a precise, documented, measurable biochemical mechanism that is nonetheless systematically ignored.
“When one of the messengers is missing, the whole system compensates, often poorly.” Dr Thierry Hertoghe
If you want to first understand the basics of thyroid function and its seven essential cofactors, start with my article on thyroid and micronutrition. Here, we’re going to talk about the deep connection between your female hormones and your thyroid, the knot that medicine never unties because it looks at each thread separately.
The Mechanism Nobody Ever Explained to You
We need to start with an invisible actor you won’t find on any prescription: TBG, thyroxin-binding globulin. It’s the transport protein for thyroid hormones in the blood. T4 and T3 don’t circulate freely. They’re bound to this protein, like passengers on a bus. And only the hormones that get off the bus, the ones called “free” (free T4, free T3), can enter cells and act.
The problem is that estrogens increase TBG production by the liver. The more estrogen in the blood, the more the liver makes TBG, and the more thyroid hormones get sequestered on their transport proteins. There’s less free T3 available for cells. Your blood test might show normal total T4, correct TSH, and yet your cells are starving because free T3 is trapped. As Hertoghe reminds us in The Hormone Handbook, excess estrogens increase TBG and reduce free thyroid hormones, creating a picture of functional hypothyroidism that TSH alone doesn’t detect.
This mechanism alone explains why women are eight times more affected than men. It’s not a weakness. It’s a direct biochemical consequence of female physiology. Every time estrogens rise, TBG rises, and available free T3 drops. And every period in a woman’s life when estrogens fluctuate dramatically becomes a period of thyroid risk.
My article on thyroid and liver details this bidirectional relationship: the liver converts 60% of T4 to T3, but it also makes TBG. When the liver is overloaded with estrogens, it produces too much TBG and no longer converts enough T3. Double trouble.
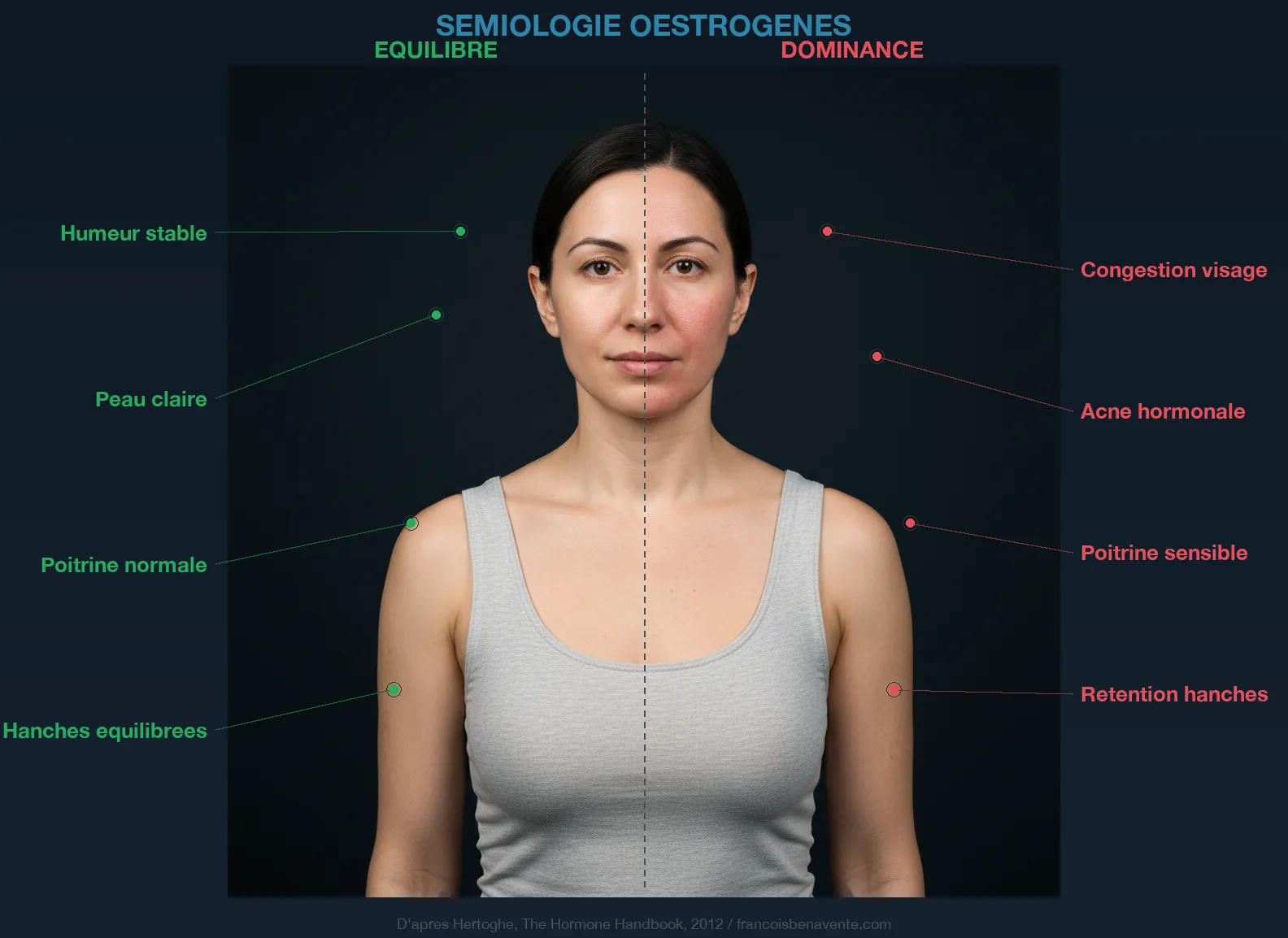
The Problem Isn’t Just Excess Estrogens
We often think of “estrogenic dominance” when discussing this imbalance. But as I explain in the article on period pain, dominance doesn’t mean excess. You can have estrogens in the low-normal range and still be in dominance if progesterone is even lower. It’s the ratio that matters, not the absolute value.
Progesterone is the natural antagonist of estrogens. It reduces estrogen receptors, decreases their activity, desensitizes receptors to aldosterone (diuretic effect), and indirectly supports the thyroid by maintaining overall hormonal balance. Hertoghe confirms: estradiol is ten times more powerful than estrone, and without sufficient progesterone to counterbalance it, it imposes its law on the entire body. Consequences: infertility, endometriosis, PCOS, fibroids, water retention, and of course, thyroid blockade via TBG.
Why does progesterone drop? The first cause is chronic stress. Progesterone shares a precursor with cortisol: pregnenolone. When the body is in permanent stress, pregnenolone is diverted toward cortisol production at the expense of progesterone. It’s the “pregnenolone theft” I explain in detail in the article on stress, cortisol and adrenals. Then come deficiencies in magnesium, lithium, zinc, copper and manganese, essential cofactors for progesterone synthesis. Extreme diets that deprive the body of cholesterol (precursor to all steroid hormones), and statins, which block HMG-CoA reductase, also deplete the raw materials. Hertoghe adds a factor I observe daily: when the thyroid and adrenals are fatigued, there’s little or no ovulation, creating an imbalance that favors cyst formation.
All these diseases, endometriosis, PCOS, fibroids, estrogen-dependent cancers, share a common denominator: an estrogen/progesterone imbalance combined with elevated fasting blood sugar. It’s a consistent clinical observation that Hertoghe and my own consultations confirm.
The Four Periods at Risk in Your Life
I think of Lea (name changed), 17 years old, brought in by her mother. Fatigue for a year, chaotic periods, acne, hair loss. The GP said “it’s adolescence.” TSH was 3.8 mU/L, “within normal range.” Free T3 wasn’t tested. When I ordered it, it was 2.6 pmol/L. Rock bottom. And nobody had connected the estrogenic storm of puberty to this thyroid quietly failing.
Four moments in a woman’s life concentrate most thyroid triggering events. These aren’t coincidences. They’re hormonal crossroads where estrogens shift, TBG follows, and the thyroid pays the price.
Puberty and the pill form the first crossroads. At puberty, estrogens explode. TBG rises. It’s when many young girls start developing diffuse thyroid symptoms nobody identifies: fatigue, weight gain, concentration problems, cold sensitivity. We blame it on adolescence. Then comes the pill, often prescribed at 15 or 16 for painful periods or acne. The synthetic estrogens in the pill increase TBG exactly like natural estrogens do. Worse: they overload the liver, which now has to metabolize synthetic molecules on top of its normal functions. T4 to T3 conversion slows. Zinc reserves collapse (Curtay’s work confirms that vitamin B6 levels drop in women on the pill for more than two years, and zinc follows the same trajectory). Magnesium plummets. And natural progesterone is put on standby since the pill suppresses ovulation.
I think of Sarah (name changed), 26 years old, who came to me after eight years on the pill. She’d stopped for a pregnancy plan, and since then everything had collapsed. Missing cycles, crushing fatigue, ten kilos in six months. Her gynecologist had suggested going back on the pill “until things settle.” Nobody had tested her free T3, zinc, magnesium, vitamin D. Nobody had looked at her liver. Her body was screaming that eight years of synthetic estrogens had emptied the reserves and blocked thyroid conversion. The article on hypothyroidism as a symptom explains why this diagnostic wandering is so frequent.
Pregnancy and postpartum form the second crossroads, and probably the most violent. During pregnancy, estrogens multiply by thirty. This is necessary for placental development, uterine vascularization, baby growth. But the thyroid consequence is massive: TBG explodes, and the mother’s available free T3 drops drastically. The baby’s body needs T3 to build its brain (one third EPA) and the myelin sheaths of its neurons (one third DHA). And as Curtay and Hertoghe clarify, T4 doesn’t cross the placenta, only T3 counts. Any maternal T3 insufficiency directly impacts fetal neurological development.
The American Thyroid Association recommends TSH below 2.5 mU/L for conception. This threshold is much stricter than typical lab ranges, and yet it’s rarely checked. One in five women who’ve had a miscarriage have a thyroid problem. And deficiencies accumulate: anemia multiplies obstetric complication risk by 2.5 to 3. Curtay reports that 80% of women are magnesium deficient (Val-de-Marne study), and 100% of women of childbearing age don’t meet their zinc needs through food. Pregnancy draws from reserves that are already empty.
After delivery, the drop in placental hormones creates abrupt hormonal void. Postpartum thyroiditis affects 5 to 10% of women. It often goes unnoticed because fatigue, mood changes and weight gain are attributed to sleep deprivation and “adjusting to motherhood.” It’s a diagnosis that should be systematically sought within six months after delivery.
Perimenopause, roughly between 40 and 50 years old, is the third crossroads. It’s the most treacherous period because hormones don’t decrease linearly. They oscillate. One cycle, estrogens are at the ceiling. The next, they’re at the floor. Progesterone drops more steadily since ovulation becomes intermittent. Result: months of intense estrogenic dominance alternate with months of global deficiency. The thyroid endures these roller coasters without a net. This is when we see late-onset Hashimoto, “new” hypothyroidism in women who never had problems. The article on Hashimoto explains how the autoimmune mechanism can trigger when terrain is weakened by this hormonal instability.
Menopause closes the cycle. Estrogens drop permanently, but the thyroid doesn’t recover. Years of estrogenic dominance have exhausted the liver, emptied cofactor reserves, and sometimes triggered an irreversible autoimmune process. Plus, the estrogen drop comes with accelerated bone loss, increased cardiovascular risk, and metabolic slowdown that worsens thyroid symptoms. It’s often at this point that Levothyrox gets prescribed, without anyone seeking to understand the decades of imbalance that led there.
Good and Bad Estrogens
Not all estrogens are equal, and this distinction is fundamental. The liver metabolizes estrogens through cytochrome P450 enzymes, and depending on the pathway, the result is radically different.
CYP1A1 and CYP3A4 enzymes hydroxylate estrogens at position 2. 2-OH metabolites have weak estrogenic activity. They’re protective, anti-proliferative, associated with lower cancer risk. This is the pathway you want to favor.
CYP1B1 hydroxylates at position 4. 4-OH metabolites are potentially genotoxic, capable of forming DNA adducts.
And then there’s the 16-alpha-OH pathway. 16-alpha-OH metabolites retain high estrogenic activity, are pro-inflammatory, and stimulate cell proliferation. And here’s the connection nobody makes: 16-alpha estrogens increase hepcidin, that hepatic hormone blocking intestinal iron absorption and sequestering it in macrophages. It’s the bridge between excess estrogens and the anemia so many women drag without explanation. Less available iron also means less functional thyroperoxidase, and therefore less thyroid hormone production.
There’s a fascinating clinical hypothesis I observe in consultations: excess 16-alpha-OH metabolites could explain the ten-women-to-one-man ratio in autoimmune thyroiditis. It’s these pro-inflammatory metabolites that maintain the cascade described by Seignalet, that xenoimmune response that ends up attacking the thyroid. Men, with estrogen levels ten to twenty times lower, are naturally protected from this mechanism.
To orient toward the protective 2-OH pathway, the liver needs cruciferous vegetables (broccoli, kale, rocket, radish, broccoli sprouts rich in sulforaphane and indole-3-carbinol), vitamin B6, magnesium, and efficient methylation (B9 as 5-MTHF, B12 methylcobalamine, betaine). DIM (diindolylmethane) is the active metabolite of indole-3-carbinol. It’s one of the supplements I prescribe most in consultations for women with an unfavorable estrogenic profile. Curtay insists on a crucial point: the form of B9 matters. 5-MTHF is the active form, not synthetic folic acid found in most prenatal supplements.
The Liver, the Obligatory Crossroads
You’ve understood: the liver is central to everything. It converts 60% of T4 to active T3. It makes TBG. It metabolizes estrogens through P450 cytochromes. It produces bile that eliminates conjugated estrogenic metabolites. And it synthesizes cholesterol, the precursor to all steroid hormones (including progesterone).
When the liver is overloaded, everything collapses at once. T4 to T3 conversion slows, as detailed in my article on thyroid and liver. TBG increases because the liver keeps producing it under the effect of estrogens it can’t eliminate anymore. Unmetabolized estrogens re-enter circulation (enterohepatic cycling). Bile production decreases, slowing transit and promoting estrogen reabsorption by the estrobolome, those gut bacteria producing beta-glucuronidase.
It’s a vicious circle I observe daily. Hypothyroidism slows liver metabolism. The congested liver no longer metabolizes estrogens. Estrogens increase TBG. Free T3 drops. Hypothyroidism worsens. The liver slows further. And round and round. Dr Mouton speaks of “fatty thyroid liver” to describe the steatosis so often accompanying chronic hypothyroidism.
Xenoestrogens add another layer. Non-organic meat, conventional milk, pesticides, plastics, conventional cosmetics, PFAS from nonstick pans, contaminated tap water. These molecules mimic estrogen action, saturate receptors, and overload an already overwhelmed liver. The EFSA (European Food Safety Authority) identified 101 pesticides out of 287 evaluated that directly affect the thyroid. That’s one third.
The Intestinal Dysfunction That Amplifies Everything
The intestine plays a role we systematically underestimate. The estrobolome, that collection of gut bacteria capable of metabolizing estrogens, can become your worst enemy. In case of dysbiosis, certain bacteria overproduce beta-glucuronidase, an enzyme that deconjugates estrogens the liver prepared for elimination. Instead of being excreted in feces, estrogens are reabsorbed and sent back into circulation. Estrogenic dominance worsens without you consuming a single endocrine disruptor.
Intestinal candida infection complicates matters. Candida albicans weakens tight junctions of the epithelium, creating intestinal permeability that lets pro-inflammatory macromolecules through. And in the context of Hashimoto, this permeability is the starting point of the autoimmune cascade described by Seignalet: antigenic peptides cross the barrier, accumulate in thyrocytes, and the immune system attacks the thyroid. Candida also captures magnesium via tricarballylate production, further depleting an already deficient organism.
Transit is fundamental. Constipation is the enemy of every woman in estrogenic dominance. Without daily transit, estrogens conjugated by the liver and excreted in bile stagnate and are reabsorbed. Hypothyroidism itself slows peristalsis. It’s another vicious circle.
The Plate Before the Pill
Marchesseau repeated it: “Don’t kill the mosquitoes, drain the swamp.” Before any supplementation, food. And in the thyroid-female hormones context, meals act on both fronts simultaneously.
Cruciferous vegetables are the first lever. Broccoli, kale, cauliflower, rocket, radish, turnip. Broccoli sprouts are the most concentrated in sulforaphane. Sulforaphane activates liver phase II enzymes (glucuronidation, sulfation) and indole-3-carbinol orients estrogens toward the protective 2-OH pathway. I prescribe it daily. The ginger-rosemary decoction in the morning combines hepatoprotective, cholagogue and anti-inflammatory effects. Ginger inhibits COX-2 with efficacy comparable to ibuprofen in clinical studies on dysmenorrhea, without the gastric side effects.
Reduce excessive omega-6 (sunflower oil, processed foods, frying) and increase omega-3 (small fatty fish three times a week, flaxseed or camelina oil in dressing). The ideal omega-6/omega-3 ratio is 1 to 3. Western diet is often 20 to 1. This imbalance fuels chronic inflammation that aggravates both estrogenic dominance and hypothyroidism.
Quality proteins, especially at breakfast: organic eggs, small fatty fish, nuts. Target: 1.2 g per kg of body weight daily. Amino acids are precursors to neurotransmitters and hormones. Tyrosine, direct precursor to thyroid hormones, comes from animal proteins.
Priority eliminations: refined sugar (insulin resistance, candida, inflammation), alcohol (direct liver overload), non-fermented soy in excess (phytoestrogens in estrogenic-dominant women), conventional dairy (xenoestrogens, pro-inflammatory casein), gluten if intestinal permeability suspected. The article on naturopathy basics details the overall dietary reform.
The Cofactors That Unlock the Situation
I think of Nathalie (name changed), 43 years old, perimenopausal. Intense fatigue for two years, weight gain of seven kilos, dry skin, chaotic periods. TSH at 3.4 mU/L, “within normal range.” Free T3 at 2.9 pmol/L. Fluctuating estradiol. Progesterone at rock bottom. Plasma zinc at 0.55 mg/L (low-normal range). Low erythrocyte magnesium. Vitamin D at 19 ng/mL. Ferritin at 25 ng/mL. Nobody had prescribed this workup. Her endocrinologist monitored TSH, her gynecologist suggested hormone replacement. Neither looked at cofactors.
Zinc first. Cofactor for T4 to T3 conversion, cofactor for delta-6-desaturase (which converts omega-3 to anti-inflammatory prostaglandins), immune modulator. According to Dr Claeys, over 90% of thyroid patients are zinc deficient. And Curtay confirms that 100% of women of childbearing age don’t meet their needs through food. Zinc bisglycinate, 15 to 25 mg daily, away from meals rich in iron and calcium.
Magnesium next. Anti-stress, muscle relaxant, cofactor for over 300 enzymatic reactions. 80% of women are deficient according to the Val-de-Marne study. Magnesium supports the adrenals (less pregnenolone theft), relaxes uterine muscle, and participates in T4 to T3 conversion. Magnesium bisglycinate or malate, 300 to 400 mg daily, in two doses.
Vitamin B6 as P5P (pyridoxal-5-phosphate, the active form). Cofactor for delta-6-desaturase, cofactor for liver detoxification of estrogens in phase II, cofactor for progesterone and serotonin synthesis. The pill consumes B6, excess estrogens consume B6, and without B6, neither omega-3 conversion nor estrogen detoxification works properly. P5P, 50 mg daily.
Selenium: cofactor for 5’-deiodinase (T4 to T3 conversion), cofactor for glutathion peroxidase (major thyroid antioxidant). Selenomethionine, 100 to 200 micrograms daily. Essential before any iodine supplementation, especially in case of Hashimoto.
Omega-3 EPA and DHA: precursors to anti-inflammatory PGE3 prostaglandins and resolvins. EPA is the direct precursor to PGE3 that relaxes uterine muscle and reduces inflammation. Fish oil or microalgae, 2 to 3 g daily.
Vitamin D3: immunomodulator, anti-inflammatory. The ENNS study shows over 80% of the French population is insufficient. Blood target: 60 ng/mL. Vitamin D3, 2,000 to 4,000 IU daily, with vitamin K2-MK7.
Iron, but only after ferritin testing. Target: 50 to 90 ng/mL. Free iron is pro-oxidant (Fenton reaction). Curtay warns: iron plus vitamin C supplementation generates hydroxyl radicals. Prefer iron combined with magnesium and polyphenols. Never supplement blindly.
Finally, B9 as 5-MTHF (never synthetic folic acid) to support liver methylation and estrogen detoxification. Homocysteine target is 7 mg/L or below. Above that, methylation is insufficient, and estrogens stagnate.
For supplementation, Sunday Natural offers pharmaceutical-quality selenium, zinc, magnesium and vitamin D (-10% with code FRANCOIS10).
The Phytotherapy Protocol by Cycle Phase
I’m drawing here on the protocol of Rina Nissim, Swiss midwife and author of Mamamelis, adapted to the thyroid-female hormones issue. The idea is to support each cycle phase with plants that support the dominant hormone of that phase.
In follicular phase (from first day of period to ovulation): blackcurrant (anti-inflammatory, adrenal support), raspberry leaf (uterine tonic, mineral-rich), horsetail (remineralizing, silicon-rich), bramble (astringent, tonic). This is when estrogens naturally rise. We don’t block them, we ensure the liver metabolizes them correctly.
In luteal phase (from ovulation to period): gromwell (restrains excessive estrogen production), lady’s mantle (the progesterone-like plant par excellence, uterine tonic), yarrow (hormonal regulator, antispasmodic), chasteberry (Vitex agnus-castus, acts on the hypothalamic-pituitary axis by favoring LH secretion, supports progesterone production by the corpus luteum). Chasteberry is the most-studied plant for cycle disorders. Nissim recommends 80 drops twice daily in tincture during the luteal phase.
Additionally, a hot water bottle on the right upper abdomen each evening, 20 minutes. This simple gesture is the most powerful way to activate liver circulation, promote bile production, and accelerate estrogen detoxification. Dr Salmanoff placed capillary therapy at the center of all regeneration: warm baths, hot water bottles, sweating techniques open emunctories and decongest the liver.
The article on period pain details Nissim’s phytotherapy protocol in full, and the one on endometriosis applies it to the specific context of that disease.
Special Case: Pregnancy and Conception
Preconception preparation is the moment when the thyroid-female hormones knot must be imperative untied. The stakes are too high to leave to chance. I’ve devoted an entire article to this topic: thyroid and fertility, the lock nobody unlocks.
The American Thyroid Association sets TSH target below 2.5 mU/L for conception. It’s a threshold many labs don’t even flag, since their ranges go up to 4.0 or 4.5. Any woman planning pregnancy should demand a complete periconception workup: TSH, free T3, free T4, anti-TPO and anti-thyroglobulin antibodies. Not just TSH.
Iodine needs increase 50% during pregnancy. Iron needs double. Zinc, selenium, magnesium and B vitamin needs increase massively. I’ve detailed periconception supplementation in a dedicated article. And the fetus depends entirely on the mother for thyroid hormones during the first trimester, before its own thyroid becomes functional. Maternal T3 deficit during this critical window has measurable consequences on the child’s IQ.
Curtay insists on B9 form: 5-MTHF, not synthetic folic acid. Some women carry the MTHFR polymorphism that prevents them from converting folic acid to its active form. They think they’re protected by their prenatal supplement, but it doesn’t work. Homocysteine is the marker to monitor. Iron must be paired with magnesium and polyphenols, never with vitamin C supplement (Fenton reaction). For baby’s brain development, omega-3 is non-negotiable: one third EPA for the brain, one third DHA for myelin sheaths.
The Workup to Demand
I recommend this workup to any woman presenting with thyroid symptoms fluctuating with the cycle, a long-term pill history, difficult postpartum, or entering perimenopause with fatigue and weight gain.
Complete thyroid workup: TSH, free T3, free T4, reverse T3, T3L/rT3 ratio, anti-TPO and anti-thyroglobulin antibodies. TSH alone isn’t enough. It doesn’t detect TBG blockade or hepatic conversion defect.
Female hormone workup: estradiol (E2), progesterone (in luteal phase, around day 21), SHBG, free testosterone, DHEAS. The E2/progesterone ratio is more informative than isolated values. If possible, urine testing of estrogenic metabolites (2-OH, 4-OH, 16-alpha-OH) lets you assess liver detoxification profile. Before blood testing, you can assess your estrogenic dominance with the Hertoghe estrogen questionnaire.
Cofactor workup: plasma zinc, erythrocyte magnesium (not serum, it’s misleading), ferritin, 25-OH-vitamin D, plasma selenium, homocysteine, vitamin B12, erythrocyte B9.
Liver and metabolic workup: gamma-GT, AST, ALT, bilirubin, triglycerides, total cholesterol and fractions, fasting glucose, fasting insulin (HOMA-IR).
This complete workup lets you visualize both systems simultaneously, thyroid and female hormonal, and the metabolic bridges linking them. It’s from this data that your protocol can be individualized. The article on thyroid and weight explains why estrogens increase receptor numbers in adipocytes and make fat stubborn.
What Naturopathy Cannot Do
I want to be clear. Naturopathy doesn’t replace endocrinological or gynecological follow-up. Serious thyroid diseases (Graves’ disease, thyroid cancers, myxedema) require medical management. Deep endometriosis, PCOS with severe insulin resistance, large fibroids, uncontrolled menorrhagia are in medicine’s domain.
Naturopathy accompanies. It identifies and corrects the terrain imbalances that maintain the thyroid-female hormones knot. It supports the liver, repairs the intestine, fills deficiencies, reduces inflammation, rebalances prostaglandins, supports progesterone. Both approaches don’t oppose. They complement, and it’s in this complementarity that the most lasting results build.
I think back to Nathalie. After five months of protocol (cofactor correction, liver support, phytotherapy by cycle phase, dietary reform), her free T3 had risen from 2.9 to 4.6 pmol/L. Her progesterone had doubled. Her cycles had stabilized. She’d lost four kilos without dieting. And most importantly, that crushing fatigue accompanying her for two years had vanished. She hadn’t taken Levothyrox. She’d untied the knot.
If you want personalized support, you can book a consultation.


Laisser un commentaire
Sois le premier à commenter cet article.